微生物作为天然产物的重要源泉,在自然界中分布广泛,包括真菌、细菌、病毒,以及一些小型的原生生物、显微藻类等生物群体,在进化过程中可以产生大量结构新颖多变的代谢产物,是一个巨大的分子宝库(张勇慧 2018)。地衣是地衣型真菌和绿藻或蓝细菌的结合,菌类从藻类获得光合作用制造的有机物,藻类则依靠菌类提供水、无机盐和二氧化碳,彼此结成特殊的互惠共生关系(彭霄鹏和温仕良 2019)。除了共生真菌伴侣外,地衣体中还存在内生真菌,它们是一大类之前被忽略的真菌类群,生活在内髓层部位,不引起明显病理症状(元超等2019)。近年来的研究显示,地衣内生真菌作为隐藏在地衣体内的微生物仓库,其发酵产物具有丰富的化学结构多样性,是新颖化合物的重要来源(Yang et al. 2018)。
轮层炭壳菌属Daldinia,隶属于子囊菌门Ascomycota,核菌纲Pyrenomycetes,炭角菌目Xylariales,炭角菌科Xylariaceae,是一类重要的木材腐朽菌,最早于1863年被发现(Cesati & De 1863)。据报道,轮层炭壳菌属Daldinia内生真菌能够产生多种结构新颖的次级代谢产物(梅荣清等 2019),例如从该属真菌中发现具有新型结构的四氢萘酮类(Liao et al. 2019)、色原酮类(Luo et al. 2018)、萘醌类(Kamauchi et al. 2018)、细胞松弛素类(Yang et al. 2018)、异香豆素类(Quang et al. 2013)等化合物,使其成为天然产物化学研究的新热点之一;但这些内生真菌大多从植物中分离得到,从地衣中分离得到轮层炭壳菌属Daldinia内生真菌的相关报道较少。
本课题组首次从一星点梅衣属地衣Punctelia sp.中分离了一株轮层炭壳菌属内生真菌蔡氏轮层炭壳菌Daldinia childiae,为了进一步挖掘D. childiae次级代谢产物,本研究对其固体发酵产物进行了初步研究,已期为开发该属菌株可能存在的应用价值奠定基础。
1 材料与方法
1.1 供试材料
1.1.1 菌株:星点梅衣属地衣Punctelia sp.于2018年3月采集自贵州省都匀市斗篷山景区,从中分离获得一株内生真菌,标本号为DPS-165-9,结合形态特征并在GenBank中通过ITS序列比对,鉴定为蔡氏轮层炭壳菌Daldinia childiae(GenBank号:MT139887)。该菌株现保存于中国科学院微生物研究所菌物保藏中心(CGMCC),保存编号CGMCC No. 17469,菌种PDA斜面保藏于4℃冰箱。
1.1.2 培养基:PDA培养基用于地衣内生真菌的分离纯化,PDB培养基用于菌株的孵育,大米固体培养基用于菌株的发酵培养。PDA培养基配方:马铃薯200g,葡萄糖20g,琼脂15g,蒸馏水1 000mL,pH自然;其配方中不加入琼脂,即得PDB培养基。
1.1.3 主要试剂和仪器:石油醚、二氯甲烷、乙酸乙酯、四氢呋喃等有机试剂(分析纯,天津富宇精细化工有限公司);GF254薄层色谱硅胶板,柱层析硅胶(100-200目,300-400目)(青岛海洋化工厂),DPPH(1,1-二苯基-2-三硝基苯肼,C18H12N5O6)和维生素C(上海源叶生物科技有限公司)。
SW-CJ-IDLRH-250型立式单人净化工作台购自苏州净化设备有限公司;TS-2102C型恒温振荡培养箱购自上海天呈科技有限公司;BXM-30R型压力蒸汽灭菌锅购自上海博迅实业有限公司医疗设备厂;EYELA-N-110型旋转蒸发仪购自日本东京理化器械株式会社;Agilent DD2400-MR型核磁共振仪购自美国Agilent公司;ZF-7型三相紫外分析仪购自上海嘉鹏科技有限公司;Agilent 6210 ESI/TOF型质谱仪购自美国Agilent公司;多功能酶标仪购自赛默飞世尔科技有限公司。
1.2 方法
1.2.1 菌株的活化与种子液的培养:将保存在PDA斜面的菌株接种至PDA平板上,25℃培养4d,活化2-3代;用接种棒将活化菌株接种于15个250mL锥形瓶中(每瓶装有100mL PDB培养基,121℃高压蒸汽灭菌20min),在室温摇床上振荡培养4d,转速150r/min,获得种子液。
1.2.2 菌株的发酵:配制大米固体培养基60瓶(500mL三角瓶,70g大米,200mL水,3.5g葡萄糖),121℃高压蒸汽灭菌20min,冷却至室温备用;在无菌条件下,每瓶接种20mL种子液,室温下静置培养30d。
1.2.3 菌株的提取与分离:将静置培养30d的菌株发酵物捣碎后,乙酸乙酯浸泡过夜,提取3次,合并滤液,减压浓缩,获得粗提物20.0g。粗提物以石油醚/乙酸乙酯系统(100/0,100/10,100/20,100/50,50/100,0/100,V/V)为溶剂进行梯度洗脱,得到6个粗组分(Fr.1-Fr.6)。Fr.1经硅胶柱层析,石油醚洗脱得化合物4(1.3g)和组分Fr.1-1,Fr.1-1采用重结晶的方法得到化合物5(1.0g);Fr.2用硅胶柱层析(石油醚:二氯甲烷=1:1,V/V)等度洗脱得化合物6(159.4mg);Fr.3经硅胶柱层析,二氯甲烷洗脱得化合物2(7mg),用硅胶柱层析(石油醚:二氯甲烷=1:1,V/V)等度洗脱得化合物3(28.1mg);Fr.4用硅胶柱层析(石油醚:乙酸乙酯=2:1,V/V)等度洗脱得化合物1(35.7mg);Fr.5用硅胶柱层析(石油醚:乙酸乙酯=3:1,V/V)等度洗脱得组分Fr.5-1,Fr.5-1再经过硅胶柱层析(石油醚:四氢呋喃=1:1,V/V)洗脱得化合物8(50.0mg);Fr.6用硅胶柱层析(石油醚:乙酸乙酯=1:1,V/V)等度洗脱得化合物Fr.6-1,Fr.6-1再经过硅胶柱层析(石油醚:四氢呋喃=1:1,V/V)洗脱得化合物7(25.0mg)。
1.2.4 自由基清除活性实验:(1)反应液的配制:称取4mg DPPH用甲醇溶解并定容于50mL容量瓶中得DPPH储备液,4℃冰箱避光保存。取DPPH储备液15mL置于25mL容量瓶,用甲醇稀释至刻度,摇匀,避光静置,得DPPH工作液。(2)样品溶液和Vc溶液的配制:用微量移液器吸取1mL甲醇溶解称量好的3mg各样品及Vc,采用二倍稀释法对初始浓度的样品和Vc进行稀释得到5个浓度梯度的样品溶液和Vc溶液。(3)吸光度值的测定:参考李媛媛等(2017)的方法,加以适当修改。在96孔板中依次加入200μL DPPH工作液和5μL不同浓度的样品溶液和Vc溶液,充分混匀,室温下测定波长517nm处的吸光度,对照组用甲醇代替样品溶液。按照计算公式:清除率(%)=[(A对照组-A样品组)/A对照组]× 100,对所得的数据进行处理分析。
2 结果与分析
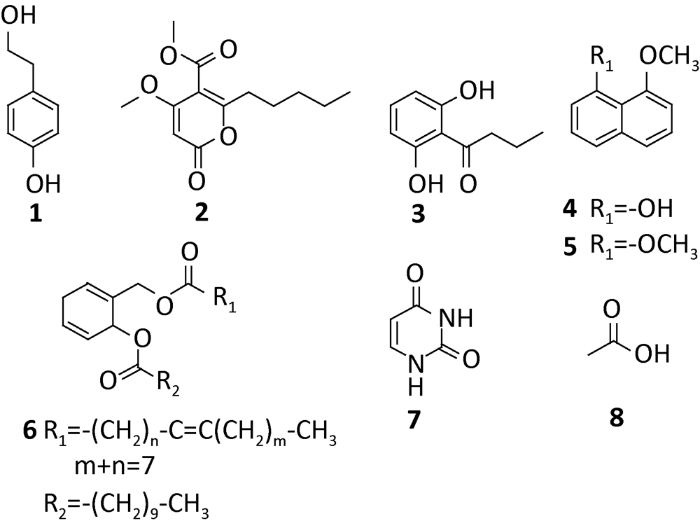
对蔡氏轮层炭壳菌Daldinia childiae进行大米固体发酵培养,通过硅胶柱色谱和重结晶的方法对乙酸乙酯提取物进行分离纯化,得到8个单体化合物(图1),分别鉴定为:4-hydroxy-benzeneethanol(1)、4-methoxy-5-car-bomethoxy-6-pentyl-2H-pyran-2-one(2)、1-(2,6- dihydroxyphenyl)butan-1- one(3)、8-methoxy-1-naphthol(4)、1,8- dimethoxynaphthalene(5)、methyl 2-undece noate-3-undecanoate--cyclohexyl-1,4-dienyl ester(6)、uracil(7)和acetic acid(8)。
图1
2.1 化合物1的结构鉴定
黄色液体;1H-NMR(400MHz,CD3OD)δ:7.01(2H,dd,J=8.4,2.0Hz,H-2′,6′),6.69(2H,dd,J=8.4,2.0Hz,H-3′,5′),3.66(2H,t,J=7.2Hz,H-1),2.72(2H,t,J=7.2Hz,H-2);13C-NMR(100MHz,CD3OD)δ:155.3(s,C-4′),131.2(s,C-1′),131.0(d,C-2′,6′),114.7(d,C-3′,5′),63.2(t,C-1),38.0(t,C-2)。以上数据与袁莉等(2016)的报道基本一致,故化合物1鉴定为4-hydroxy- benzeneethanol。
2.2 化合物2的结构鉴定
黄色油状;HR-ESI-MS m/z 255.1232[M+H]+,277.1052[M+Na]+。1H-NMR(400MHz,CDCl3)δ:5.43(1H,s,H-3),3.81(3H,s,4-OCH3),3.83(3H,s,12-OCH3),2.50(2H,t,J=7.6Hz,H-7),1.67(4H,dd,J=7.2,4.4Hz,H-8,H-9),1.29(2H,m,H-10),0.86(3H,t,H-11);13C-NMR(100MHz,CDCl3)δ:168.3(C-4),167.0(C-6),164.5(C-12),163.1(C-2),108.8(C-3),87.9(C-5),56.7(4-OCH3),52.9(12-OCH3),32.6(C-7),31.4(C-8),27.2(C-9),22.4(C-10),14.1(C-11);HMBC谱中,H-7与C-6相关,-OCH3与C-4相关,-OCH3与C-12相关,以上数据与Quang et al.(2002)的报道基本一致,故化合物2鉴定为4-methoxy-5-car-bomethoxy-6-pentyl-2H-pyran- 2-one。
2.3 化合物3的结构鉴定
黄色粉末;1H-NMR(400MHz,CDCl3)δ:9.80(2H,s,Ar-OH)6.38(2H,d,J=8.0Hz,H-4,H-6),7.22(1H,dd,H-5),3.10(2H,t,J=7.2Hz,H-2′),1.79(2H,m,J=7.2Hz,H-3′),0.98(3H,t,H-4′);13C-NMR(100MHz,CDCl3)δ:207.9(C-1′),161.2(C-1,C-3),135.7(C-5),110.0(C-2),108.3(C-4,C-6),46.7(C-2′),17.8(C-3′),13.9(C-4′)。以上数据与Dai et al.(2006)的报道基本一致,故化合物3鉴定为1-(2,6-dihydroxyphenyl)butan- 1-one。
2.4 化合物4的结构鉴定
白色固体;1H-NMR(400MHz,CDCl3)δ:9.37(1H,s,1-OH),7.41(1H,d,J=8.3Hz,H-4),7.36-7.28(3H,m,H-3,H-5,H-6),6.95(1H,d,J=7.5Hz,H-7),6.63(1H,d,J=8.0Hz,H-2),4.13(3H,s,8-OCH3);13C-NMR(100MHz,CDCl3)δ:156.0(C-8),154.5(C-1),136.7(C-4a),127.6(C-3),125.6(C-6),121.6(C-5),118.7(C-4),114.9(C-8a),110.3(C-2),103.7(C-7),55.7(8-OCH3)。以上数据与李冬利等(2011)的报道基本一致,故化合物4鉴定为8-methoxy- 1-naphthol。
2.5 化合物5的结构鉴定
无色针晶;1H-NMR(400MHz,CDCl3)δ:7.37(2H,dd,J=8.2,1.4Hz,H-4,H-5),7.35(2H,dd,J=8.2,8.2Hz,H-3,H-6),6.84(2H,dd,J=8.2,1.4Hz,H-2,H-7),3.97(6H,s,1-OCH3,8-OCH3);13C-NMR(100MHz,CDCl3)δ:157.3(C-1,C-8),137.6(C-4a),126.5(C-3,C-6),121.0(C-4,C-5),117.8(C-8a),106.5(C-2,C-7),56.6(1-OCH3,8-OCH3)。以上数据与黄华容等(2005)的报道基本一致,故化合物5鉴定为1,8-dimethoxynaphthalene。
2.6 化合物6的结构鉴定
淡绿色油状;HR-ESI-MS m/z 459.2049[M-H]-。1H-NMR(400MHz,CDCl3)δ:5.28(5H,m,H-1,H-4,H-5,H-9′′,H-10′′),5.21(1H,s,H-3),4.25(2H,dd,J=7.6Hz,H-1′′′),1.24-2.24(34H,m,17×CH2),0.85(6H,m,10′′-CH3,10′-CH3);13C-NMR(100MHz,CDCl3)δ:173.2(C-1′′),172.8(C-1′),128.1-130.0(C-1,C-2,C-4,C-5,C-9′′,C-10′′),69.0(C-1′′′),62.2(C-3),22.6-34.0(17C,17×CH2),14.1(10′′-CH3,10′-CH3)。结合李千会和徐德平等(2018)的描述,化合物6鉴定为methyl 2-undecenoate-3-undecanoate- cyclohexyl-1,4-dienyl ester。
2.7 化合物7的结构鉴定
白色结晶;1H-NMR(400MHz,DMSO-d6)δ:10.93(1H,s,H-1),10.72(1H,s,H-3),5.35(1H,d,J=7.6Hz,H-5),7.38(1H,d,J=7.6Hz,H-6);13C-NMR(100MHz,DMSO-d6)δ:164.4(C-4),151.6(C-2),142.3(C-6),100.6(C-5)。以上数据与Wei et al.(2017)的报道基本一致,故化合物7鉴定为uracil。
2.8 化合物8的结构鉴定
白色结晶;1H-NMR(400MHz,DMSO-d6)δ:12.06(1H,s,-OH),2.33(3H,s,-CH3);13C-NMR(100MHz,DMSO-d6)δ:174.0(C-1),29.1(C-2)。结合以上数据及王素娟和裴月湖(2000)的描述,化合物8鉴定为acetic acid。
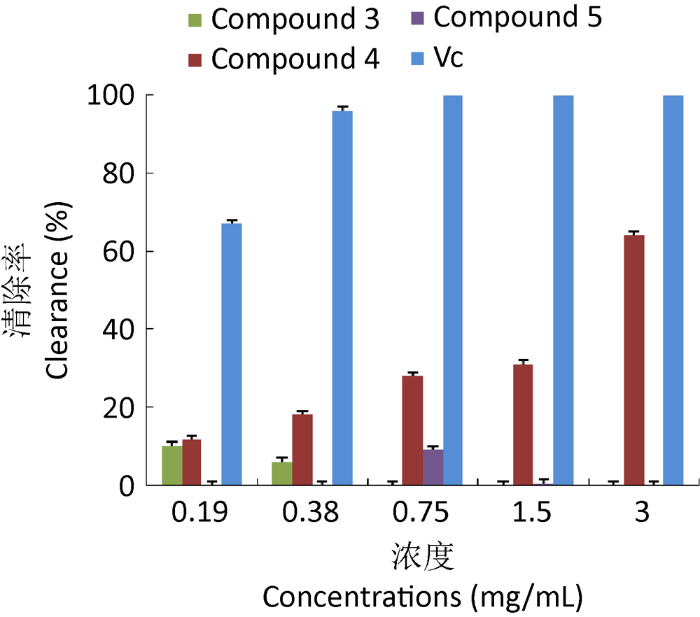
2.9 自由基清除活性实验
通过抗氧化活性实验发现,化合物3,4,5对DPPH自由基有一定的清除能力。化合物4和Vc的浓度越高,对DPPH自由基的清除率越大,即清除能力越强,且影响显著;当质量浓度为3.0mg/mL时,化合物4对DPPH自由基的清除率达到最大值,为64%,可能与结构中的酚羟基有关,化合物3和化合物5对DPPH自由基的清除率相对较弱(图2)。
图2
3 讨论
微生物是先导化合物的重要来源,真菌是微生物的重要类群之一,种类繁多,形态各异,生活环境多样,真菌中分离得到的代谢产物也多种多样(雷红梅和赵沛基 2019)。但是从常规真菌资源中筛选新化合物面临着巨大的挑战,而那些生长在自然环境恶劣、营养条件匮乏地区的特殊生境真菌为了生存,会逐步形成不同于普通土壤真菌的独特生存策略,进而代谢结构新颖多样的化学成分,地衣内生真菌就具有这种特殊性(李晓彬 2015)。地衣内生真菌与地衣的组织器官等具有紧密的关联,生存方式特别,生境特殊,作为潜在的巨大新型物质资源,对其中的新颖结构代谢产物进行挖掘具有重要的意义(解斐和娄红祥 2013)。
通过对一株地衣内生真菌蔡氏轮层炭壳菌Daldinia childiae固体发酵产物的化学成分进行研究,分离得到8个单体化合物,结构鉴定为4-hydroxy-benzeneethanol(1)、4-methoxy-5-car-bomethoxy-6-pentyl-2H-pyran-2-one(2)、1-(2,6-dihydroxyphenyl)butan- 1-one(3)、8-methoxy-1-naphthol(4)、1,8-dimethoxynaphthalene(5)、methyl 2-undecenoate-3-undecanoate-cyclohexyl- 1,4-dienyl ester(6)、uracil(7)和acetic acid(8);其中化合物1,6和8均首次从轮层炭壳菌属Daldinia中分离得到;并研究了化合物3,4,5的自由基清除活性,发现化合物4在浓度为3mg/mL时对DPPH自由基的清除率达到64%,具有较强的自由基清除活性,化合物3和化合物5对DPPH自由基的清除率相对较弱,具有轻微的自由基清除能力;研究结果为进一步挖掘轮层炭壳菌属的活性代谢产物奠定基础,同时丰富了对地衣内生真菌化学成分的认识。
参考文献
Schema di classificazione degli sferiacei italici aschigeri piu’o meno appartenenti al genere Sphaeria nelľ antico significato attribuitoglide Persoon
Metabolites from the endophytic fungus Nodulisporium sp. from Juniperus cedre
Metabolites of mangrove endophytic fungus 3920 from the South China Sea
Isoindolinones, phthalides and a naphthoquinone from the fruiting body of Daldinia concentrica
DOI:10.1021/acs.jnatprod.7b00976
URL
PMID:29715021
[本文引用: 1]

A chemical investigation of the ascomycetes of Daldinia concentrica was performed using silica gel column chromatography, ODS column chromatography, and preparative HPLC. Two new isoindolinone compounds, daldinans B (1) and C (2), two new phthalide compounds, daldinolides A (3) and B (4), and a new naphthoquinone, daldiquinone (5), were isolated together with two known compounds (6 and 7). The structures of 1, 2, and 5 were established using NMR, MS, and IR methods, and the structures of 3 and 4 were determined by derivatization from known compounds (6 and 7). 5 exhibited antiangiogenesis activity against HUVECs (IC50 = 7.5 muM).
Advances in research on mining fungal secondary metabolites
Chemical constituents of endophytic fungus Nodulisporium sp. A4 from Aquilaria sinensis
OBJECTIVE: To investigate the chemical constituents of an endophytic fungus, Nodulisporium sp. A4, from the medicinal plant Aquilaria sinensis and search for antitumor natural products. METHOD: The fungus was cultured in liquid medium and extracted with EtOAc. The compounds were isolated by various chromatographic methods (silica gel, reverse silica gel, Sephadex-LH20, preparative TLC and so on) and recrystallization. Structural elucidation was conducted by extensive analysis of spectroscopic data as well as by comparison with literature reports. The antitumor activity of isolated compounds was tested by MTT method in vitro. RESULT: Seven compounds were isolated and identified from the broth culture, their structures were determined to be 5-methyl-2-vinyltetrahydrofuran-3-ol (1), 6-methyl-2-(5-methyl-5-vinyltetrahydrofuran-2-yl) hept-5-en-2-ol (2), 6alpha-hydroxycyclonerolidol (3), rel-(1S,4S, 5R,7R,10R)-10-desmethyl-1-methyl-11-eudesmene (4), tyrosol (5), 8-methoxynaphthalen-1-ol (6), and 1,8-dimethoxynaphthalene (7). Three compounds were isolated and identified from the mycelia as ergosterol (8), ergosterol peroxide (9), and cerevisterol (10). The in vitro pharmalogical evaluation results displayed that compounds 3 and 4 showed 89.1%, 44.2% and 82.3%, 79.8% inhibition against tumor cell lines SF268 and NCI-H460 at 100 mg x L(-1), respectively. CONCLUSION: Compound 1 was a new natural product, compounds 2, 3, 7 and 10 were reported from the genus Nodulisporium sp. for the first time. Compounds 3 and 4 exhibited weak inhibitory effects on the proliferation of tumor cell lines SF268 and NCI-H460.
Chemical constituents of pumpkin seeds against benign prostatic hyperplasia
Study on chemical components and biological activity of five endophytic fungi
A micro-model of DPPH, ABTS and FRAP for dynamic monitoring of the antioxidant activity of different types of polyphenols isolated from red wine
Bioactive polyketide derivatives from the mangrove- derived fungus Daldinia eschscholtzii HJ004
DOI:10.1021/acs.jnatprod.9b00241 URL [本文引用: 1]
A new chromone and a new aliphatic ester isolated from Daldinia eschscholtzii
Studies on bioactive secondary metabolites from a mangrove-derived fungus Daldinia eschscholtzii HJ00
Lichens: the symbiosis of bacteria and algae
Chemical constituents of the ascomycete Daldinia concentrica
DOI:10.1021/np020301h URL [本文引用: 1]
Cytotoxic constituents from the fungus Daldinia concentrica (Xylariaceae)
DOI:10.1080/14786419.2012.698411 URL [本文引用: 1]
Studies on the chemical constituents of the leaves of Betula platyphylla Suk
α-Glucosidase inhibitors and phytotoxins from Streptomyces xanthophaeus
DOI:10.1080/14786419.2016.1269100
URL
PMID:28013556
[本文引用: 1]

Twenty-four metabolites 1-24 were isolated from the fermentation broth of Streptomyces xanthophaeus. Their structures were elucidated on the basis of spectroscopic analysis and by comparison of their NMR data with literature data reported. Daidzein (1), genistein (2) and gliricidin (3) inhibited alpha-glucosidase in vitro with IC50 values of 174.2, 36.1 and 47.4 muM, respectively, more potent than the positive control, acarbose. Docking study revealed that the amino acid residue Thr 215 is the essential binding site for active ligands 2. In addition, the phytotoxic effects of all compounds were assayed on radish seedlings, five of which, 3, 8, 13, 15 and 18, inhibited the growth of radish (Raphanus sativus) seedlings with inhibitory rates of >60% at a concentration of 100 ppm, which was comparable or superior to the positive control glyphosate. This is the first report of the phytotoxicity of the compounds.
Research progress on endolichenic fungi and their active secondary metabolites
New phomalone derivatives from the endolichenic fungus Cochliobolus kusanoi in Ny-Alesund Arcti
DOI:10.1002/jccs.2019.66.issue-3 URL [本文引用: 1]
One new cytochalasin metabolite isolated from a mangrove-derived fungus Daldinia eschscholtzii HJ001
DOI:10.1080/14786419.2017.1346641
URL
PMID:28658974
[本文引用: 1]

One new cytochalasin metabolite [11]-cytochalasa-5(6),13-diene-1,21-dione-7,18-dihydroxy-16,18-dimethyl-10-phenyl-(7S*,13E,16S*,18R*) (1), together with three known compounds (2-4) were obtained from the EtOAc extract of the endophytic fungus Daldinia eschscholtzii HJ001 isolated from the mangrove Brguiera sexangula var. rhynchopetala collected in the South China Sea. Their structures were elucidated by the detailed analysis of comprehensive spectroscopic data. Compounds 1 and 2 were evaluated for their antibacterial and cytotoxic activities.
Secondary metabolites from Epicoccum nigrum 14 one, an endophytic fungus isolated from plant Leptogium masiaticum
Chemical investigation on the South China Sea sponge Iotrocota sp
Study on active natural products from microorganisms
南海红树林内生真菌3920代谢产物研究
对采自香港的红树林内生真菌(3920号)的代谢产物进行了研究,从中分离得到4个化合物.其中1,8-二甲氧基萘和1-(2,6-二羟基)苯丁酮首次从海洋真菌中分离得到.它们的结构通过FABMS,NMR和X-单晶数据分析得到确定.
白木香内生真菌多节孢Nodulisporium sp. A4的化学成分研究
微型DPPH、ABTS和FRAP法动态监测从红酒中分离得到的不同种类多酚化合物的抗氧化活性
一株红树来源真菌Daldinia eschscholtzii HJ001的活性次级代谢产物研究
白桦叶化学成分的研究
从白桦(Betula platyphylla Suk.)的叶中分离得到9个化合物,根据理化性质和波谱数据鉴定为邻二苯酚(1),对羟基苯甲醛(2),对二苯酚(3),3,4-二羟基苯甲酸(4),3-甲氧基-4-羟基苯甲酸(5),2-呋喃甲酸(6),没食子酸(7),丁二酸(8),β-谷甾醇(9).
一株植物内生真菌Epicoccum nigrum 14 one次生代谢产物研究