植物内生真菌(endophytic fungi)是指某阶段生活在健康植物组织或器官内部,与植物形成特殊的相互关系(Jia et al. 2016),对宿主不会引起明显病害的真菌(孙剑秋等 2008;Aschehoug et al. 2012;Kusari et al. 2012)。据报道,几乎所有植物体内均含有内生真菌,且两者相互依存(Aly et al. 2011)。植物在一定程度上可调节内生真菌的代谢过程,如产生对微生物和宿主具有保护作用的化合物(Kusari et al. 2012)。同时,内生真菌也能通过调节植物体内的激素代谢、增强对营养元素的吸收促进宿主植物生长(Lugtenberg & Kamilova 2009;Jia et al. 2016);或通过分泌他感物质提高药用植物在群落中的竞争能力(Ahlholm et al. 2002)。近期的研究指出,药用植物微生物很可能是宿主健康和生产力的重要决定因素(Rana et al. 2019;Dong et al. 2020)。大多药用植物内生真菌可以产生和宿主植物相同或相似的代谢产物(陈晓芳等 2020)。如炭疽菌Colletotrichum spp.产生的青蒿素和安德鲁紫杉菌Taxomyces andreanae产生的紫杉醇(Huang et al. 2018)。内生真菌的定殖率、多样性和群落组成受宿主基因型、组织类型和非生物因素的影响(Compant et al. 2019;Li et al. 2020)。如,Li et al.(2020)研究发现,10种荒漠盐生植物根内生真菌的定殖率和物种丰富度各不相同。大针茅Stipa grandis根系内生真菌的定殖率显著高于叶片,根和叶中内生菌的多样性和组成具有明显差异(Su et al. 2010)。尽管植物根和叶中内生菌的研究取得了重要进展,但树皮仍是一个缺乏探索的微生境(Chen et al. 2018;Pellitier et al. 2019)。此外,有研究指出不同地理来源的同一物种也会因所处环境的不同导致植物内生微生物存在差异(Lau et al. 2013)。例如,Sun et al.(2020)研究表明环境影响了肉苁蓉Cistanche deserticola土壤微生物的组成和功能,从而导致药用植物的差异化。因此,分析不同地理区域药用植物树皮内生真菌群落的组成、多样性及差异将为揭示其形成的驱动因素和生态功能等方面提供重要科学资料。
杜仲Eucommia ulmoides Oliver是杜仲科Eucommiaceae杜仲属Eucommia的落叶乔木,属我国特有经济树种和滋补药材,广泛分布于我国湘西北、黔北、黔西及陕南等地(周政贤和郭光典 1980)。不同产地的杜仲药材品质存在差异,可能受气候因子、温度、湿度、海拔及土壤微生物等影响,同时受到自身内部微环境的作用(杨娟等 2019)。杜仲内生真菌是构成植物内环境的重要部分,对杜仲植物的生长、次级代谢产物合成及药用成分有重要影响(Lu et al. 2000;Chen et al. 2010)。相关研究表明,杜仲内生真菌发酵提取物中能检测到与宿主具相同药效的松脂醇二葡萄糖苷、绿原酸等活性成分,这些成分是评价杜仲药材质量的重要指标(Chen et al. 2010;Ding et al. 2013;梁雪娟等 2014)。我们前期研究发现,部分树皮微生物与杜仲药理活性成分呈正相关性,内生真菌群落可影响杜仲活性成分的合成(董醇波等 2019;杨娟等 2019;Dong et al. 2020)。然而,目前的研究报道主要是针对单一或少数地区的杜仲内生真菌进行了分离鉴定、多样性及其代谢产物功能评价,缺少对不同环境、多省区的杜仲内生真菌多样性的全面解析,影响了对跨区域杜仲内生真菌种类的深入认识和其进一步的功能研究。因此,大尺度、多途径对杜仲不同部位的内生真菌多样性和群落组成研究具有重要意义。
杜仲Eucommia ulmoides Oliver是杜仲科Eucommiaceae杜仲属Eucommia的落叶乔木,属我国特有经济树种和滋补药材,广泛分布于我国湘西北、黔北、黔西及陕南等地(周政贤和郭光典 1980)。不同产地的杜仲药材品质存在差异,可能受气候因子、温度、湿度、海拔及土壤微生物等影响,同时受到自身内部微环境的作用(杨娟等 2019)。杜仲内生真菌是构成植物内环境的重要部分,对杜仲植物的生长、次级代谢产物合成及药用成分有重要影响(Lu et al. 2000;Chen et al. 2010)。相关研究表明,杜仲内生真菌发酵提取物中能检测到与宿主具相同药效的松脂醇二葡萄糖苷、绿原酸等活性成分,这些成分是评价杜仲药材质量的重要指标(Chen et al. 2010;Ding et al. 2013;梁雪娟等 2014)。我们前期研究发现,部分树皮微生物与杜仲药理活性成分呈正相关性,内生真菌群落可影响杜仲活性成分的合成(董醇波等 2019;杨娟等 2019;Dong et al. 2020)。然而,目前的研究报道主要是针对单一或少数地区的杜仲内生真菌进行了分离鉴定、多样性及其代谢产物功能评价,缺少对不同环境、多省区的杜仲内生真菌多样性的全面解析,影响了对跨区域杜仲内生真菌种类的深入认识和其进一步的功能研究。因此,大尺度、多途径对杜仲不同部位的内生真菌多样性和群落组成研究具有重要意义。
以湖南、四川、贵州、陕西、河南、安徽、江西7个省的杜仲树皮为研究对象,分析杜仲树皮可培养内生真菌群落组成及多样性,为后续杜仲的深入研究及生产实践中有益内生真菌的开发利用提供理论依据和后备资源。
1 材料与方法
1.1 材料
杜仲树皮样品分别采自湖南慈利县、四川旺苍县、贵州遵义市、河南洛阳市、安徽亳州市、江西赣州和陕西略阳7个地区,样本编号分别为HU、SC、GZ、HN、AH、JX和SX。在各采集位点随机挑选3-5棵7-8年长势健康的杜仲树,在离地1-1.5m高处,利用采集工具刮取适量的新鲜树皮,然后将其装入编号的无菌保鲜袋中(采集工具必须进行灭菌处理),带回实验室放于4℃条件保存,备用。
1.2 方法
1.2.1 消毒处理:采用经典三步消毒法(王红霞等 2020)(乙醇-次氯酸钠-乙醇)对采集的杜仲树皮进行消毒,具体步骤如下:用无菌水冲洗杜仲树皮的表面杂质,于超净工作台用无菌刀将杜仲树皮切块,再用无菌滤纸将表面的水吸干,将样品置于75%无水乙醇中浸泡2min,再用无菌水冲洗2-3遍,随后放入5%次氯酸钠溶液中浸泡3-5min,取出继续用75%无水乙醇消毒30s,最后使用无菌水将表面残留的乙醇溶液冲洗干净,用无菌滤纸擦干,备用。
1.2.2 培养基制备:利用马铃薯葡萄糖琼脂培养基(PDA)对杜仲树皮可培养内生真菌进行分离纯化。PDA培养基制备如下:马铃薯洗净去皮,称取200g切成小块,加水煮烂(约20min),用8层纱布过滤,滤液中加入18-20g琼脂,加热搅拌、溶解完后,加入20g葡萄糖,搅拌均匀,用蒸馏水定容至1 000mL,pH自然,在121℃条件下灭菌30min(张亮等 2017)。
1.2.3 内生真菌的分离、纯化及保种:用无菌刀刮掉杜仲树皮表面的栓皮层,将中心韧皮部切割成0.2cm×0.2cm的小块,再将其接种至PDA培养基,每个培养基放5小块(每个处理3个重复)。置于25℃恒温培养箱中培养3-5d。待菌丝长出,用接菌针从边缘挑取菌丝并移至新的PDA平板上,采用菌丝顶端纯化法对分离出的菌种进行逐步纯化。将纯化好的菌株接到PDA斜面上,25℃培养4-5d,放于4℃冰箱保藏备用(杨娟等 2019)。
1.3 杜仲树皮内生真菌的形态学鉴定
1.4 杜仲树皮内生真菌的分子鉴定
1.4.1 DNA的提取:参照真菌基因组DNA提取试剂盒(北京百泰克生物技术有限公司)进行DNA提取。
1.4.2 PCR扩增反应:采用引物ITS1:(5°-TCC GTAGGTGAACCTGCGG-3°)与ITS4:(5°-TCCTC CGCTTATTGATATGC-3°)。PCR反应条件:94℃预变性5min;94℃变性1min,50℃退火1min,72℃延伸1min,35个循环;72℃延伸10min,PCR反应体系(25µL):ddH2O 8.5µL,模板2µL,引物ITS1和ITS4均为1µL,master-mix为12.5µL。通过琼脂糖凝胶电泳检测PCR扩增产物,合格后送至昆明硕擎生物科技有限公司进行测序。
1.5 序列数据分析
对测序获得的待鉴定菌株的ITS序列,经手工校对。登陆NCBI(
1.6 杜仲内生真菌多样性研究
1.6.1 分离频率:采用分离频率(isolation frequency,IF)来比较判断不同地区杜仲树皮内生真菌的优势菌群。分离频率是指某一指定类型(某一属)内生真菌分离到的菌株数与分离出的总内生真菌菌株数的比值。计算公式如下:
$ IF=\frac{某一属内生真菌的菌株数}{分离出的总内生真菌株数}\times 100 \%$
1.6.2 多样性指数:是指用简单的数值表示群落内种类多样性的程度,用来判断群落或生态系统的稳定性指标。Shannon-Wiener多样性指数(H′)和Simpson优势度指数(D):利用Shannon-Wiener多样性指数(H′)、Simpson优势度指数(D)来评估不同产地杜仲树皮内生真菌群落多样性。H′指数越大,说明相应的内生真菌群落多样性越大,群落复杂程度越高;D指数正好相反,其指数越大,相应内生真菌群落多样性越小,群落复杂程度越低。
Shannon-Wiener多样性指数(H′)公式:
H′=-$~\underset{i=1}{\overset{n}{\mathop \sum }}\,$(Pi)(InPi),Pi是指某一内生真菌占全部内生真菌总数的百分数。
Simpson(D)指数公式:D=-$~\underset{i=1}{\overset{S}{\mathop \sum }}\,$(Pi)2,S
为物种总数,Pi为属于属/种i的个体在全部个体中的比例(杨娟等 2019)。
1.6.3 丰富度和均匀度:Magalef即是群落丰富度,Magalef指数越高,物种越丰富。Pielou 均匀度指数是指某一群落或生境中全部物种个体数目的分配状况,其反映了各物种个体数目分配的均匀程度。
Margalef丰富度指数(R)计算公式:R=(S-1)/lnN,式中S表示物种数,N表示样本总个体数。
Pielou均匀度指数(J)计算公式:J=H′/lnS,H′为Shannon-Wiener指数,S表示物种数。
1.6.4 Jaccard相似性系数(Cj):Jaccard(Cj)相似性系数主要用于比较两样本之间真菌种类组成的相似程度。公式:Cj=j/(a+b-j),式中j为两样品间共有种数或属数;a和b分别为样品A和样品B的物种数或属数。
2结果与分析
2.1 杜仲树皮可培养内生真菌菌群组成
从7个样地杜仲树皮中共分离到545株内生真菌,鉴定为7纲、15目、27科、43属(表1)。在纲水平上,主要隶属于散囊菌纲Eurotiomycetes、粪壳菌纲Sordariomycetes、座囊菌纲Dothideomycetes、伞菌纲Agaricomycetes、毛霉纲Mucoromycetes、锤舌菌纲Leotiomycetes及伞形菌纲Umbelopsidomycetes。其中以粪壳菌纲为优势纲,共分离到259株,占菌株总数的46.79%,其次是散囊菌纲,共分离到149株,占比为27.34%。在目水平上,优势目为散囊菌目Eurotiales和间座壳目Diaporthales,分别占菌株总数的27.34%和22.02%。在属水平上,以青霉属Penicillium(22.39%)和间座壳属Diaporthe(16.7%)为优势属,随后为镰刀菌属Fusarium(7.89%)、链格孢属Alternaria(4.77%)、曲霉属Aspergillus(4.59%)和枝孢属Cladosporium(4.04%)。上述结果表明,杜仲树皮中具有丰富多样的内生真菌。
表1 杜仲树皮可培养真菌的菌群组成
Table 1
| 纲 Class | 目 Order | 科 Family | 属 Genus | 菌株数 Number | 分离频率 IF (%) |
|---|---|---|---|---|---|
| 散囊菌纲 | 散囊菌目 | 曲霉科 | 曲霉属 | 25 | 4.59 |
| Eurotiomycetes | Eurotiales | Aspergillaceae | Aspergillus | ||
| 青霉属 | 122 | 22.39 | |||
| Penicillium | |||||
| 发菌科 | 蓝状菌属 | 2 | 0.37 | ||
| Trichocomaceae | Talaromyces | ||||
| 粪壳菌纲 | 间座壳目 | 间座壳科 | 间座壳属 | 91 | 16.7 |
| Sordariomycetes | Diaporthales | Diaporthaceae | Diaporthe | ||
| 黑腐皮壳科 | 拟茎点霉属 | 26 | 4.77 | ||
| Valsaceae | Phomopsis | ||||
| 小圆锥孢菌属 | 3 | 0.55 | |||
| Schizoparmaceae | Coniella | ||||
| 假毛球壳目 | 假毛球壳科 | 黑孢属 | 8 | 1.47 | |
| Trichosphaeriales | Trichosphaeriaceae | Nigrospora | |||
| 肉座菌目 | 丛赤壳科 | 镰刀菌属 | 43 | 7.89 | |
| Hypocreales | Nectriaceae | Fusarium | |||
| Thyronectria | 9 | 1.65 | |||
| Cylindrocladiella | 1 | 0.18 | |||
| 赤霉属 | 3 | 0.55 | |||
| Gibberella | |||||
| 肉座菌科 | 木霉属 | 16 | 2.94 | ||
| Hypocreaceae | Trichoderma | ||||
| 枝顶孢霉属 | 3 | 0.55 | |||
| Acremonium | |||||
| 生赤壳科 | 枝穗霉属 | ||||
| Bionectriaceae | Clonostachys | 18 | 3.3 | ||
| 生赤壳属 | 1 | 0.18 | |||
| Bionectria | |||||
| 炭角菌目 | 炭角菌科 | 中碳垫菌属 | 1 | 0.18 | |
| Xylariales | Xylariaceae | Nemania | |||
| 炭角菌属 | 1 | 0.18 | |||
| Xylaria | |||||
| 荚胞腔菌科 | 拟盘多毛孢属 | 15 | 2.75 | ||
| Sporocadaceae | Pestalotiopsis | ||||
| 炭角菌科 | 炭团菌属 | 6 | 1.1 | ||
| Hypoxylaceae | Hypoxylon | ||||
| 粪壳菌目 | 毛壳菌科 | 毛壳菌属 | |||
| Sordariales | Chaetomiaceae | Chaetomium | 8 | 1.47 | |
| 腐质霉属 | 6 | 1.1 | |||
| Humicola | |||||
| 刺盘孢目 | 半边莲科 | ||||
| Glomerellales | Plectosphaerellaceae | Plectosphaerella | 4 | 0.73 | |
| 座囊菌纲 | 葡萄座腔菌目 | 葡萄座腔菌科 | 葡萄座腔菌属 | 11 | 2.02 |
| Dothideomycetes | Botryosphaeriales | Botryosphaeriaceae | Botryosphaeria | ||
| 毛双孢菌属 | 7 | 1.28 | |||
| Lasiodiplodia | |||||
| 穴壳菌属 | 3 | 0.55 | |||
| Dothiorella | |||||
| Neofusicoccum | 1 | 0.18 | |||
| 枝孢霉目 | 枝孢霉科 | 枝孢属 | 22 | 4.04 | |
| Cladosporiales | Cladosporiaceae | Cladosporium | |||
| 格孢菌目 | 亚隔孢壳科 | 附球菌属 | 2 | 0.37 | |
| Pleosporales | Didymellaceae | Epicoccum | |||
| 亚隔孢壳属 | 7 | 1.28 | |||
| Didymella | |||||
| Paracamarosporium | 3 | 0.55 | |||
| 假性小毛球菌属 | 6 | 1.1 | |||
| Incertae sedis | Pseudochaetosphaeronema | ||||
| 格孢菌科 | 链格孢属 | 26 | 4.77 | ||
| Pleosporaceae | Alternaria | ||||
| 小光壳属 | 1 | 0.18 | |||
| Leptosphaerulina | |||||
| 孢黑团壳科 | 长蠕孢属 | 11 | 2.02 | ||
| Massarinaceae | Helminthosporium | ||||
| Medicopsis | 2 | 0.37 | |||
| 多孔菌目 | 原毛平革菌科 | 烟管菌属 | 1 | 0.18 | |
| Polyporales | Phanerochaetaceae | Bjerkandera | |||
| 伞菌纲 | 木耳菌目 | 木耳科 | 木耳属 | 2 | 0.37 |
| Agaricomycetes | Auriculariales | Auriculariaceae | Auricularia | ||
| 毛霉纲 | 毛霉菌目 | 小克银汉霉科 | 犁头霉属 | 1 | 0.18 |
| Mucoromycetes | Mucorales | Cunninghamellaceae | Absidia | ||
| 小克银汉霉属 | 6 | 1.1 | |||
| Cunninghamella | |||||
| Gongronella | 15 | 2.75 | |||
| 毛霉菌科 | 毛霉属 | 3 | 0.55 | ||
| Mucoraceae | Mucor | ||||
| 锤舌菌纲 | 柔膜菌目 | 暗色孢科 | |||
| Leotiomycetes | Helotiales | Dermateaceae | Cryptosporiopsis | 1 | 0.18 |
| 伞形霉纲 | 伞形霉目 | 伞形霉科 | 伞形霉属 | 8 | 1.47 |
| Umbelopsidomycetes | Umbelopsidales | Umbelopsidaceae | Umbelopsis |
2.2不同产地杜仲树皮内生真菌群落组成差异
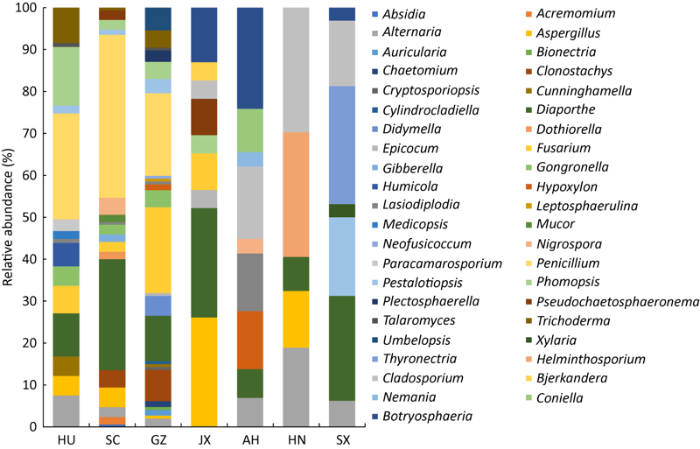
2.2.1 不同产地杜仲树皮内生真菌的优势类群、常见类群和稀有类群:依据庞雄飞和尤民生(1996)对优势属、常见属和稀有属的定义:某菌群占整体菌群的百分比≥10%,即为优势属,1%-10%时为常见属,≤1%为稀有属。在湖南样本中鉴定出15属,共107株,分离频率为19.63%,其中青霉属(25.23%)和拟茎点霉属Phomopsis(14.01%)为优势属,蓝状菌属Talaromyces(0.93%)和毛双孢菌属Lasiodiplodia(0.93%)为稀有属,其余11个属为常见属;四川样本分离到18属,共170株,分离频率为31.19%,青霉属(38.82%)和间座壳属(26.47%)是优势属,犁头霉属Absidia(0.59%)、赤霉属Gibberella(0.55%)、小光壳属Lasiodiplodia(0.59%)和木霉属Trichoderma(0.59%)为稀有属,其余12个属为常见属;贵州样本中内生真菌隶属于25属,共147株,分离频率26.97%,优势属有镰刀菌属(20.41%)、青霉属(19.73%)和间座壳属(10.88%),常见属有11属,稀有属有10属;江西样本和安徽样本均获得为9属,江西样本23株,安徽样本29株,分离频率分别为4.22%和5.32%,江西样本优势属是曲霉属和间座壳属,均占26.09%,其次是葡萄座腔菌属Botryosphaeria(13.04%),其余为常见属;安徽样本优势属为葡萄座腔菌属(24.14%)、枝孢属(17.24%)、炭团菌属Hypoxylon(13.79%)、小光壳属(13.79%)和小圆锥孢菌属Coniella(10.34%),其余为常见属;河南样本的37株菌株归属于5个属,分离频率为6.79%,优势属为枝孢属和长蠕孢属Helminthosporium,均为29.73%,其次是链格孢属(18.92%)、曲霉属(13.51%)和间座壳属(8.11%)为常见属;陕西样本鉴定出7属,共32株,分离频率5.87%,Thyronectria(28.13%)、间座壳属(25.00%)、拟盘多毛孢属Pestalotiopsis(18.75%)和枝孢属(15.63%)为优势属,其余为常见属。这些结果表明,在不同地区的杜仲树皮中内生真菌菌群组成和数量都具有一定差异(图1)。
图1
图1
在属水平上不同产地真菌群落组成和相对多度 HU:样本来自湖南慈利县;SC:样本来自四川旺苍县;GZ:样本来自贵州遵义市;HN:样本来自河南洛阳市;AH:样本来自安徽亳州市;JX:样本来自江西赣州市;SX:样本来自陕西略阳县. 下同
Fig. 1
Different fungal community composition and relative abundance at the generic level. HU: The sample was from Cili County, Hunan Province; SC: The sample was from Wangcang County, Sichuan Province; GZ: The sample was from Zunyi, Guizhou Province; HN: The Sample was from Luoyang, Henan Province; AH: The Sample was from Bozhou, Anhui Province; JX: The Sample was from Ganzhou, Jiangxi Province; SX: The sample was from Lueyang County, Shaanxi Province. The same below.
2.2.2 不同产地杜仲树皮内生真菌多样性分析:Shannon指数越大,说明对应样品中的真菌群落多样性越大;Simpson指数正好相反,指数越大,对应样品中的真菌群落多样性越小(杨娟等 2019)。Magalef指数是群落丰富度,Magalef指数越高,则说明该群落越丰富;Pielou是均匀度指数,Pielou指数越高,则说明该群落越均匀。杜仲树皮内生真菌多样性指数见表2。Shannon多样性指数(H′)从高到低依次为四川(3.73)>湖南(3.41)>安徽(2.93)>贵州(2.88)>江西(2.79)>河南(2.18);Simpson优势度指数(D)从大到小依次为贵州(0.89)>湖南(0.88)>安徽(0.85)>江西(0.82)>陕西(0.79)>四川(0.77)>河南(0.76);Magalef丰富度(R)分别为贵州(24.80)>四川(17.81)>湖南(14.99)>安徽(8.70)>江西(8.68)>陕西(6.71)>河南(4.72);Pielou均匀度(J)则为河南(1.35)>安徽(1.33)>四川(1.29)>江西(1.27)>湖南(1.26)=陕西(1.26)>贵州(0.89)。Shannon指数分析可知,四川样本中真菌群落多样性最大,河南样本中真菌群落多样性最小;Simpson指数显示各样本间差异不大,贵州样本中内生真菌类群优势度最大,河南样本优势度最小;Magalef丰富度和Pielou均匀度表明在河南样本真菌群落分布最均匀,丰富度最低;贵州样本真菌群落丰富度最高,但最不均匀。该结果表明不同产地杜仲树皮内生真菌的菌落组成、丰度及多样性不同。
表2 不同产地杜仲树皮可培养内生真菌菌群的多样性
Table 2
| 样地 Sample sites | 真菌属数 No. of genera | 多样性指数 H′ | 优势度指数 D | 丰富度指数 R | 均匀度指数 J |
|---|---|---|---|---|---|
| HU | 15 | 3.41 | 0.88 | 14.99 | 1.26 |
| SC | 18 | 3.73 | 0.77 | 17.81 | 1.29 |
| GZ | 25 | 2.88 | 0.89 | 24.8 | 0.89 |
| JX | 9 | 2.79 | 0.82 | 8.68 | 1.27 |
| AH | 9 | 2.93 | 0.85 | 8.7 | 1.33 |
| HN | 5 | 2.18 | 0.76 | 4.72 | 1.35 |
| SX | 7 | 2.45 | 0.79 | 6.71 | 1.26 |
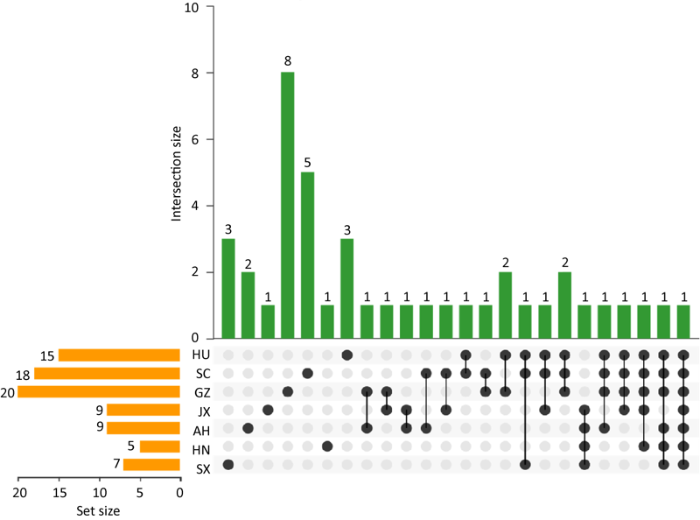
2.2.3 不同产地杜仲树皮内生真菌的共有属和特有属:从各产地真菌群落的Venn图分析可知(图2),从湖南、四川、贵州、江西、安徽、河南、陕西产地各分离出菌株15、18、25、9、9、5、7株,共有属仅有1个,即间座壳属。除此之外,一些产地间有其他相同的真菌组成,如湖南和贵州的共有属有小克银汉霉属Cunninghamella和蓝状菌属。湖南、四川及贵州的共有属为Gongronella和木霉属。同时一些真菌呈现出一定特异性和专一性,在不同样地都有各自特有类群。其中贵州样本中的特有属最多,达8个,分别是木耳属Auricularia、生赤壳属Bionectria、枝孢属、Cryptosporiopsis、Cylindrocladiella、亚隔孢壳属Didymella、小光壳属和Neofusicoccum。其次是四川样本含有5个特有属,分别为犁头霉属、枝顶孢霉属Acremonium、穴壳菌属Dothiorella、赤霉属和毛霉属Mucor。湖南有腐质霉属Humicola、Medicopsis和Paracamarosporium共3个特有属。陕西样本的3个特有属为炭角菌属Xylaria、葡萄座腔菌属和Thyronectria。安徽样本中则为碳垫菌属Nemania和小圆锥孢菌属。江西和河南的特有属都为1个,分别是长蠕孢属和烟管菌属Bjerkandera。综上所述,不同产地杜仲皮内生真菌群落组成与分布存在较明显差异(表2)。
图2
图2
在属水平上各样本真菌群落韦恩图
Fig. 2
Venn diagram of fungal community in each sample at the generic level.
2.3 不同产地杜仲树皮内生真菌的相似性
相似性系数是反映两个不同生境中真菌菌群组成相似程度的重要参数。Jaccard相似性系数原理为:当Cj为0.00-0.25时,为极不相似;Cj为0.25-0.50时,为中等不相似;Cj为0.50-0.75时,为中等相似;当Cj为0.75-1.00时为极相似。不同产地之间的杜仲树皮可培养真菌组成的相似性见表4,结果显示不同产地之间的内生真菌类群的相似性系数处于0.1111-0.4348,其中样本四川-湖南、四川-贵州、湖南-贵州、陕西-安徽、陕西-河南之间的相似性系数分别为0.4348、0.4286、0.3438、0.3333、0.3333,均属中等不相似;其余样本间相似性系数都处于0.00-0.25,为极不相似。整体来看,样本之间相似性系数都小于0.5,属于不同程度不相似,说明不同产地间杜仲树皮内生真菌组成结构不尽相同。
表3 不同产地杜仲树皮可培养真菌相似性系数比较
Table 3
| 样点 Sample sites | HU | SC | GZ | JX | AH | HN | SX |
|---|---|---|---|---|---|---|---|
| HU | 1 | 0.4348 | 0.4286 | 0.2 | 0.1429 | 0.1765 | 0.1579 |
| SC | 1 | 0.3438 | 0.2273 | 0.1739 | 0.15 | 0.1364 | |
| GZ | 1 | 0.1724 | 0.1333 | 0.1111 | 0.1034 | ||
| JX | 1 | 0.2 | 0.2727 | 0.2308 | |||
| AH | 1 | 0.2727 | 0.3333 | ||||
| HN | 1 | 0.3333 | |||||
| SX | 1 |
3 讨论
本研究从7个产地共分离到545株真菌,经形态学和分子生物学鉴定为43属。主要优势类群为青霉属(22.39%)和间座壳属(16.7%)。其中,间座壳属为湖南、四川、贵州、河南、安徽、江西、陕西7个产地的共有属。该结果与早前研究报道有相似之处,如梁雪娟(2014)揭示拟茎点霉属、间座壳属和链格孢属为慈利、略阳和遵义产地杜仲树皮内生真菌的共有属和优势属。同时,杨娟等(2019)发现贵州遵义县、湖南慈利县和四川旺苍县3个产地的杜仲树皮内生真菌的共有属为拟茎点霉属、链格孢属和间座壳属,且优势属为间座壳属。综上表明,间座壳属等常见菌属广泛分布于杜仲树皮中。研究指出,药用植物的微生物组对宿主生长、生产力及道地性有重要影响(Köberl et al. 2013;Turner et al. 2013),特别是宿主的核心微生物组在其中发挥关键性作用(Chen et al. 2018;董醇波等 2019)。董醇波等(2019)揭示了杜仲树皮中的11个核心真菌属,包含本研究分离到的毛壳菌属、镰刀菌属、间座壳属和曲霉属等,这些真菌可能与宿主紧密互作,发挥重要的生物学功能。间座壳属与青霉属广泛存在于植物各组织部位中,在植物-微生物互作过程中扮演重要角色。如青霉属作为腐生真菌,能分泌抑菌活性物质、分解有机物和促进宿主对营养元素的吸收。此外,青霉属还可作为一种重要的生防菌,对大部分植物病原菌都具有拮抗作用(袁秀英等 2006;安婧婧2016)。同时,间座壳属真菌在一些植物中常以优势种群存在(Botella & Diez 2011),能产生多种蛋白酶和次级代谢产物,在农业和医学方面都具有广阔的发展前景(Dai et al. 2005;龙慧等 2020)。
多样性指数和相似性系数均说明7个产地杜仲树皮中的真菌群落结构和分布存在差异。大量研究表明,植物内生真菌的丰度、多样性和物种组成与温度、湿度、光照、地理位置、周围植被等宿主生境条件密切相关(Suryanarayanan et al. 2005;Hoffman & Arnold et al. 2008;Jia et al. 2016)。Glynou et al.(2016)揭示了不同生境中植物根部内生真菌的丰富度和多样性与降水相关的各因素呈较强的负相关关系。此外,因植物内生真菌主要来源于土壤,土壤pH、营养物质组成等的变化也可能影响杜仲内生真菌的分布(梁雪娟等 2014)。药用植物内生真菌具有一定区域特异性,与其他区域的同种植物相比,同一区域植物微生物种类的相似度更高,这是经过长期的自然选择而形成的微生态地域特征(Jiang et al. 1994;U’Ren et al. 2012;孙一帆等 2020)。因此,不同产地特有的地理环境塑造了植物体内独特的菌群组成和结构特征,正是这些特定的微生物组成赋予了中药材独有的品质特点。
内生真菌的传播遵循垂直和水平两种途径。其中,水平传播主要通过土壤或空气传播的孢子发生(Carroll 1988),这种方式倾向于环境对内生真菌分布格局的影响。而垂直传播是指内生真菌通过种子从母体植物向后代传播的方式(Rana et al. 2019)。有研究表明,植物内生菌的定殖更多受到植物遗传特性的作用。例如,垂直传播的内生真菌对羽茅Achnatherum sibiricum生长和生理特征的益处高于水平传播的内生真菌(贾彤等 2015)。植物基因型可通过影响植物的生理特
征来影响微生物组成结构,从而对植物生长、健康和生产力产生深远影响(Chen et al. 2010;Lamit et al. 2014;Edwards et al. 2015)。窄叶杨树的基因型能够影响其嫩枝的内生真菌的丰度、群落组成及共生现象,且这种由宿主基因型导致的内生菌群落组成的变化与植物生长特性密切相关(Lamil et al. 2014)。同时,Li et al.(2021)报道了植物基因型导致宿主生理特征(如植株鲜重、根代谢和叶绿素含量)的变化,从而驱动特定叶际微生物群的积累,影响叶际微生物群落组成。综上,内生真菌的定殖和种类不仅受到环境干扰,同时受植物基因型的作用(Lluvia et al. 2015;马昭等 2015;Sunil et al. 2015)。Saikkonen et al.(2004)认为内生真菌的适应性在很大程度上取决于宿主药用植物的适应性,这表明宿主植物在一定程度上决定内生真菌的定居和分布。因此,植物宿主基因型对不同产地杜仲内生真菌的分布与定殖可能具有一定影响。
参考文献
Vertically transmitted fungal endophytes: different responses of host‐parasite systems to environmental conditions
DOI:10.1034/j.1600-0706.2002.990118.x URL [本文引用: 1]
Fungal endophytes: unique plant inhabitants with great promises
DOI:10.1007/s00253-011-3270-y URL [本文引用: 1]
Classification of Penicillium Link in Ningxia and screening for biocontrol of potato nevus
Fungal endophytes directly increase the competitive effects of an invasive forb
Competitive outcomes among plants can vary in different abiotic and biotic conditions. Here we tested the effects of two phylotypes of Alternaria endophytes on the growth, competitive effects, and competitive responses of the exotic invasive forb Centaurea stoebe. Centaurea stoebe was a better competitor against North American grass species than grasses from its European home range in the absence of endophytes. However, one endophyte both increased the biomass of C. stoebe and reduced the competitive effect of North American grasses on C. stoebe. The competitive effects of C. stoebe on grass species native to North America were enhanced by both fungal endophytes, but not for native European grasses. We do not know the mechanism by which endophytes increased C. stoebe's competitive ability, and particularly against biogeographically new neighbors, but one endophyte increased the competitive ability of C. stoebe without increasing its size, suggesting mechanisms unrelated to increased growth. We tested only a fraction of the different endophytic fungi that have been found in C. stoebe, only scratching the surface of understanding their indirect effects. However, our results are the first to demonstrate such effects of a fungal endophyte infecting an invasive forb, and one of the few to show that endophyte effects on competition do not have to be mediated through herbivory.
Phylogenic diversity of fungal endophytes in Spanish stands of Pinus halepensis
DOI:10.1007/s13225-010-0061-1 URL [本文引用: 1]
Fungal endophytes in stems and leaves: from latent pathogen to mutualistic symbiont
DOI:10.2307/1943154 URL [本文引用: 1]
Core microbiome of medicinal plant Salvia miltiorrhiza seed: a rich reservoir of beneficial microbes for secondary metabolism?
DOI:10.3390/ijms19030672 URL [本文引用: 2]
Studies on a chlorogenic acid-producing endophytic fungi isolated from Eucommia ulmoides Oliver
Research and application of chemical control technology in Soybean planting
A review on the plant microbiome: ecology, functions, and emerging trends in microbial application
DOI:10.1016/j.jare.2019.03.004 URL [本文引用: 1]
Novel highly substituted biraryl ethers, phomosines D-G, isolated from the endophytic fungus Phomopsis sp. from Adenocarpus foliolosus
DOI:10.1002/(ISSN)1099-0690 URL [本文引用: 1]
Studies on some active components and antimicrobial activities of the fermentation broth of endophytic fungi DZY16 Isolated from Eucommia ulmoides Oliv
Core microbiome of fungi in the bark of medicinal plant Eucommia ulmoides
Structure and function of bacterial microbiota in Eucommia ulmoides bark
DOI:10.1007/s00284-020-02157-2 URL [本文引用: 3]
Structure, variation, and assembly of the root-associated microbiomes of rice
The local environment determines the assembly of root endophytic fungi at a continental scale
DOI:10.1111/1462-2920.13112 URL [本文引用: 1]
Geographic locality and host identity shape fungal endophyte communities in cupressaceous trees
DOI:10.1016/j.mycres.2007.10.014 URL [本文引用: 1]
Roles of plant- associated microbiota in traditional herbal medicine
DOI:10.1016/j.tplants.2018.05.003 URL [本文引用: 1]
A friendly relationship between endophytic fungi and medicinal plants: a systematic review
Effects of different transmission modes of endophytic fungal infection on the physiology and ecology of Pterygopteris chinensis
DOI:10.17521/cjpe.2015.0008 URL
Ecological distribution and elicitor activities of endophytic fungi in Changium smyrnioides
The microbiome of medicinal plants: diversity and importance for plant growth, quality and health
Chemical ecology of endophyticfungi: origins of secondary metabolites
DOI:10.1016/j.chembiol.2012.06.004 URL [本文引用: 2]
Tree genotype and genetically based growth traits structure twig endophyte communities
DOI:10.3732/ajb.1400034 URL [本文引用: 2]
Factors influencing communities of foliar fungal endophytes in riparian woody plants
DOI:10.1016/j.funeco.2013.06.003 URL [本文引用: 1]
Diversity and community of culturable endophytic fungi from stems and roots of desert halophytes in northwest China
DOI:10.3897/mycokeys.62.38923 URL [本文引用: 2]
Phyllosphere bacterial assemblage is affected by plant genotypes and growth stages
DOI:10.1016/j.micres.2021.126743 URL [本文引用: 1]
Comparative study on distribution of endophytic fungi in Eucommia barks from different habitats
Impact of seasonal changes on fungal diversity of a semi-arid ecosystem revealed by 454 pyrosequencing
The phylogenetic study of Diaporthe in southwest China
New bioactive metabolites produced by Colletotrichum sp., an endophytic fungus in Artemisia annua
DOI:10.1016/S0168-9452(99)00199-5 URL [本文引用: 2]
Plant-growth- promoting rhizobacteria
DOI:10.1146/annurev.micro.62.081307.162918
PMID:19575558
[本文引用: 1]

Several microbes promote plant growth, and many microbial products that stimulate plant growth have been marketed. In this review we restrict ourselves to bacteria that are derived from and exert this effect on the root. Such bacteria are generally designated as PGPR (plant-growth-promoting rhizobacteria). The beneficial effects of these rhizobacteria on plant growth can be direct or indirect. This review begins with describing the conditions under which bacteria live in the rhizosphere. To exert their beneficial effects, bacteria usually must colonize the root surface efficiently. Therefore, bacterial traits required for root colonization are subsequently described. Finally, several mechanisms by which microbes can act beneficially on plant growth are described. Examples of direct plant growth promotion that are discussed include (a) biofertilization, (b) stimulation of root growth, (c) rhizoremediation, and (d) plant stress control. Mechanisms of biological control by which rhizobacteria can promote plant growth indirectly, i.e., by reducing the level of disease, include antibiosis, induction of systemic resistance, and competition for nutrients and niches.
The relationship between endophytes and host plants: implications for research on geoherbalism
Insect community ecology
Environmental filtering structures fungal endophyte communities in tree bark
DOI:10.1111/mec.15237
PMID:31495020
[本文引用: 1]

The factors that control the assembly and composition of endophyte communities across plant hosts remains poorly understood. This is especially true for endophyte communities inhabiting inner tree bark, one of the least studied components of the plant microbiome. Here, we test the hypothesis that bark of different tree species acts as an environmental filter structuring endophyte communities, as well as the alternative hypothesis, that bark acts as a passive reservoir that accumulates a diverse assemblage of spores and latent fungal life stages. We develop a means of extracting high-quality DNA from surface sterilized tree bark to compile the first culture-independent study of inner bark fungal communities. We sampled a total of 120 trees, spanning five dominant overstorey species across multiple sites in a mixed temperate hardwood forest. We find that each of the five tree species harbour unique assemblages of inner bark fungi and that angiosperm and gymnosperm hosts harbour significantly different fungal communities. Chemical components of tree bark (pH, total phenolic content) structure some of the differences detected among fungal communities residing in particular tree species. Inner bark fungal communities were highly diverse (mean of 117-171 operational taxonomic units per tree) and dominated by a range of Ascomycete fungi living asymptomatically as putative endophytes. Together, our evidence supports the hypothesis that tree bark acts as an environmental filter structuring inner bark fungal communities. The role of these potentially ubiquitous and plant-specific fungal communities remains uncertain and merits further study.© 2019 John Wiley & Sons Ltd.
Biodiversity of endophytic fungi from diverse niches and their biotechnological applications. In: Singh BP (ed.) Advances in endophytic fungal research.
Evolution of endophyte-plant symbioses
Comparative study on distribution of endophytic ningpoensis from different habitats fungi in Scrophularia
Response of endophytic fungi of Stipa grandis to experimental plant function group removal in Inner Mongolia steppe, China
Diversity and ecological distribution of endophytic fungi in medicinal plants
Regulatory relationship between quality variation and environment of Cistanche deserticola in three ecotypes based on soil microbiome analysis
DOI:10.1038/s41598-019-56847-4 URL [本文引用: 1]
Isolation, identification and diversity comparison of endophytic fungi from Glycyrrhiza uralensis in two different habitats
Temporal variation of Bistorta vivipara-associated ectomycorrhizal fungal communities in the high arctic
DOI:10.1111/mec.13458 URL [本文引用: 1]
Endophytic fungi associated with cacti in Arizona
DOI:10.1017/S0953756205002753 URL [本文引用: 1]
Host and geographic structure of endophytic and endolichenic fungi at a continental scale
DOI:10.3732/ajb.1100459
PMID:22539507
[本文引用: 1]

Endophytic and endolichenic fungi occur in healthy tissues of plants and lichens, respectively, playing potentially important roles in the ecology and evolution of their hosts. However, previous sampling has not comprehensively evaluated the biotic, biogeographic, and abiotic factors that structure their communities.Using molecular data we examined the diversity, composition, and distributions of 4154 endophytic and endolichenic Ascomycota cultured from replicate surveys of ca. 20 plant and lichen species in each of five North American sites (Madrean coniferous forest, Arizona; montane semideciduous forest, North Carolina; scrub forest, Florida; Beringian tundra and forest, western Alaska; subalpine tundra, eastern central Alaska).Endolichenic fungi were more abundant and diverse per host species than endophytes, but communities of endophytes were more diverse overall, reflecting high diversity in mosses and lycophytes. Endophytes of vascular plants were largely distinct from fungal communities that inhabit mosses and lichens. Fungi from closely related hosts from different regions were similar in higher taxonomy, but differed at shallow taxonomic levels. These differences reflected climate factors more strongly than geographic distance alone.Our study provides a first evaluation of endophytic and endolichenic fungal associations with their hosts at a continental scale. Both plants and lichens harbor abundant and diverse fungal communities whose incidence, diversity, and composition reflect the interplay of climatic patterns, geographic separation, host type, and host lineage. Although culture-free methods will inform future work, our study sets the stage for empirical assessments of ecological specificity, metabolic capability, and comparative genomics.
Diversity of culturable endophytic fungi from Broussonetia papyrifera
Handbook of fungal identification
Analyses on fungal community composition of invertebrate communities Eucommia ulmoides rhizosphere soil in different areas
Isolation of endophytes and screen of antagonistic strains in poplar trees
Diversity of endophytic fungi isolated from Eucommia ulmoides and their antifungal activity against phytopathogenic fungi
Types, distribution and introduction of Eucommia ulmoides in China
不同传播方式的内生真菌感染对羽茅的生理生态影响
DOI:10.17521/cjpe.2015.0008
[本文引用: 1]

通过田间试验, 研究了垂直传播的Neotyphodium属内生真菌和水平传播的Epichloë属内生真菌对羽茅(Achnatherum sibiricum)生理生态的影响。结果表明: Neotyphodium属内生真菌感染对羽茅的株高和叶长有显著的正效应, 而Epichloë属内生真菌感染对羽茅株高和叶长的增益作用较少。感染Neotyphodium属内生真菌的羽茅最大净光合速率显著高于感染Epichloë属内生真菌的羽茅, Neotyphodium属内生真菌显著提升了羽茅的气孔限制值和水分利用效率, 而实验测定的第一年结果显示: Epichloë属内生真菌对羽茅的各个光合指标具有明显的负效应(胞间CO<sub>2</sub>浓度除外)。Neotyphodium属内生真菌感染使得宿主植物积累的可溶性糖含量显著高于感染Epichloë属内生真菌的羽茅和不染菌的羽茅植株。感染Epichloë属内生真菌的羽茅中氮含量显著高于感染Neotyphodium属内生真菌的羽茅。总之, 内生真菌的传播方式是影响羽茅生理生态学特性的一个因素, 并且垂直传播的内生真菌对羽茅生长和生理特性的益处高于水平传播的内生真菌。
2种不同生境甘草内生真菌的分离鉴定及多样性比较