Among the three accepted subgenera of Lactarius Pers., the limit of L. subg. Plinthogalus (Burl.) Hesler & A.H. Sm. is invariably clear and repeatedly confirmed by DNA sequence data whereas those of L. subg. Lactarius and L. subg. Russularia (Fr.) Kauffman seem ambiguous (Verbeken et al. 2014; Wisitrassameewong et al. 2016; Lee et al. 2019). This is in line with the unique morphological characters of L. subg. Plinthogalus: dull-colored basidiocarps, dry and (sub-) velvety pileus, white latex often changing to or staining context pinkish or pale brownish, pileipellis a trichoepithelium or hymenoepithelium often with brownish intracellular pigmentation and macrocystidia hardly present (Hesler & Smith 1979; Heilmann- Clausen et al. 1998; Le et al. 2007; Stubbe et al. 2008; Stubbe et al. 2012; Lee et al. 2019).
Species of Lactarius subg. Plinthogalus have white latex. Those with ochraceous latex, e.g. L. ochrogalactus Hashiya and L. pertersenii Hesler & A.H. Sm. are proved to be members of Lactifluus and have been transferred to that genus (Stubbe et al. 2012). When exposed to air, the latex often discolors pinkish, vinaceous-brown or pale brownish, less often to violet or lilac (Hesler & Smith 1979; Verbeken & Horak 2000; Le et al. 2007; Lee et al. 2019). Species with yellowing latex are much fewer. In North America there are L. subvernalis Hesler & A.H. Sm., L. subtomentosus Berk. & Rav. and L. xanthydrorheus Singer, but the latter two are suspected to be member of Lactifluus (Stubbe et al. 2010, 2012). Stubbe et al. (2007) and Stubbe et al. (2008) added four species with yellowing latex from Malaysia: L. flavorosescens D. Stubbe & Verbeken, L. pallidior D. Stubbe & Verbeken, L. lazulinus D. Stubbe et al. and L. mirabilis D. Stubbe et al.
One of the authors, X.H. Wang, found two specimens with typical yellowing latex in an oak forest in Henan Province in 2010. During 2017 to 2020, more than 20 collections were obtained from similar habitats in Hubei and Hunan provinces by the other authors. DNA sequences and morphologic characters prove that they are conspecific. Although there is no available DNA data for the North American and Malaysian species with yellowing latex mentioned above, the key morphological characters of the Chinese specimens firm without argument that they are a species different from other species previously reported.
1 MATERIALS AND METHODS
1.1 Morphological observation
Descriptions of sporocarps were from fresh material and micro-morphological study was performed on dried material. Basidiospores were observed and measured in Melzer’s reagent in side view, excluding ornamentation and apiculus. 20 spores were measured for each individual. The other micro-morphological structures were revived in 10% KOH then mounted with Congo-red (aqueous reagent). Pigmentation was observed with water. All drawings except those of the basidiospores were made with the aid of a drawing tube installed on a Nikon E400 microscope. Vertical sections of the pileipellis were cut approximately at the half radius of the pileus. Sections through the stipitipellis were taken from the middle part along the stipe length. Drawings of basidiospores were made by hand, using a 5 000× amplification. Measurements (and Q values) of spores are given as (a) b‒m‒c (d), in which “a” is the lowest value, “d” the highest, “m” the mean and “b‒c” covers a minimum of 90% of the values. “Q” stands for the ratio of length and width of a spore and “Q±av” for the average Q of all spores ± sample standard deviation. [n/m/p] means n spores from m basidiocarps of p specimens. Voucher specimens are deposited in the Cryptogamic section, Herbarium of Kunming Institute of Botany, Chinese Academy of Sciences (HKAS section, KUN), Fungarium of Guangdong Institute of Microbiology (GDGM), China and Mycological Herbarium of Hunan Normal University, China (MHHNU).
1.2 DNA extraction, PCR amplifications and sequencing
DNA extraction and PCR protocols followed Wang et al. (2012). Complete ITS region (c. 700bp) and part of nucLSU (c. 900bp) were amplified using primer pairs ITS1F + ITS4 or ITS5 + ITS4 and LROR + LR5 respectively (White et al. 1990; Moncalvo et al. 2000). The reason that we selected these two loci is that they have been used for species delimitation of L. subg. Plinthogalus from Europe, North America and Asia and are proved to be able to distinguish most species (Le et al. 2007; Stubbe & Verbeken 2012; Shi et al. 2018; Wang et al. 2018a; Lee et al. 2019). PCR products were visualized via UV light after electrophoresis on 1% agarose gels stained with ethidium bromide. Successful PCR products were sent to Sangon Biotech Limited Company (Shanghai, China) for Sanger sequencing using the same primers as PCR. When sequences have heterozygous INDELs or ambiguous sites, samples were sequenced bidirectionally to make contigs of the amplified regions or verify the ambiguous sites. We used the method of Hughes et al. (2013) to determine the INDELs heterozygosity and phase the heterozygous sequences into two haplotypes. Raw sequences were assembled with Sequencher v4.1.4 (Gene Codes Corporation, Ann Arbor) and consensus sequences are deposited in GenBank with accessions MW669857-MW669909 (ITS) and MW669910-MW669942 (LSU) (Figs. 1, 2). For samples with two copies differing in INDELs, both copies were submitted to GenBank and the samples received two GenBank accessions.
1.3 Taxon sampling and phylogenetic analysis
All specimens of the new species were used in the ITS analysis except for HKAS1126557 (from Chongqing) contaminated by some unknown fungus. Ten specimens covering the ITS variation were selected for the LSU analysis. Besides the DNA sequences generated in this study, three categories of DNA data were included in the analysis: 1) Hits from BLASTn: The generated ITS sequences (with the 18S and 28S ends trimmed) were submitted to BLASTn to find matches with similarity ≥92% and query cover >90% in GenBank, using default parameters. The LSU of these samples, if available, were also retrieved and included. 2) ITS and LSU sequences of all agaricoid species of L. subg. Plinthogalus found in Asia, both published in the previous work and generated by this study; 3) ITS and LSU sequences of the look-alikes, if not covered by the first two categories, i.e. those of L. pterosporus Romagn., L. romagnesi Bon, and L. ruginosus Romagn. and L. subplinthogalus Coker. These three species resemble the target species in the brown pileus, high spore ornamentation and distant lamellae (Heilmann-Clausen et al. 1998).
Alignments were made using the online version of the multiple sequence alignment program MAFFT v7 (Katoh & Toh 2008), applying the L-INS-I strategy, and were manually adjusted in BioEdit v.7.1.3.0 (Hall 1999). ITS and LSU datasets were made and analyzed separately. No partitioning was set for the ITS dataset, considering relatively low variation among the species of L. subg. Plinthogalus. Due to the high similarity among sequences, in the partial worldwide phylogeny of L. subg. Plinthogalus (Verbeken et al. 2014; Lee et al. 2019) and north temperate phylogeny given by Stubbe & Verbeken (2012), Maximum Likelihood and Bayesian Inference methods were not able to give a resolved topology with satisfactory supports for the backbones and some species. Therefore we used the time-saving Neighbor- Joining method implemented in MEGA X (Kumar et al. 2018) to infer the phylogenetic
relationships, using the following settings: bootstrap method (with 1 000 replicates) as test of phylogeny, p-distance as substitution model, transitions and transversions both included for substitutions, uniform rates among sites, homogenous pattern among lineages and pairwise deletion for gaps treatment. For the ITS dataset, L. torminosus (Schaeff.) Pers., a species of L. subg. Lactarius, was used to root the tree and for LSU we used Lactifluus volemus (Fr.) Kuntze. A NJ bootstrap proportion (NJ-BP) ≥70% was considered as significant support for a node to be monophyletic. Data matrices and trees are available at TreeBASE under S27833.
2 RESULTS
2.1 Taxon sampling and DNA characters
Four hits of ITS sequences were obtained from GenBank using BLASTn. These data involved one Thai species L. crassiusculus H.T. Le & D. Stubbe and one Australian hypogeous sample/taxon of Zelleromyces. Published ITS and LSU (if available) sequences of 19 Asian species were included: L. albidocinereus X.H. Wang et al., L. alutaceus H. Lee et al., L. atromarginatus Verbeken & E. Horak (=L. lavandulus H.T. Le & D. Stubbe), L. aurantiacopallens H. Lee et al., L. chiangmaiensis (H.T. Le & D. Stubbe) Montoya et al., L. crassiusculus H.T. Le & D. Stubbe, L. cucurbitoides H. Lee & Y.W. Lim, L. dirkii P. Uniyal et al., L. ferruginascens K. Das et al., L. friabilis H.T. Le & D. Stubbe, L. fulvescens H. Lee et al., L. incarnatus H. Lee et al., L. montoyae K. Das & J.R. Sharma, L. nigricans G.S. Wang & L.H. Qiu, L. oomsisiensis Verbeken & Halling, L. pleuromacrocystidiatus P. Uniyal et al., L. roseoligalus X.H. Wang (=L. alboroseus H. Lee et al.), L. ustulatus H. Lee et al. and an unnamed taxon “L. sp4” in Lee et al. (2019). These sequences were published in Le et al. (2007), Stubbe & Verbeken (2012), Lee et al. (2015), Hyde et al. (2016), Das et al. (2017), Shi et al. (2018), Lee et al. (2019), Wang et al. (2019) and Uniyal et al. (2019). In addition, 27 ITS and 18 LSU sequences generated from 18 Chinese specimens of L. albidocinereus, L. atromarginatus, L. ferruginascens, L. fuliginosus, L. lignyotus, L. nigricans and L. oomsisiensis by this study were included.
Among the 22 specimens of the new species sequenced, 19 specimens have ITS region without INDELs and four with one INDEL. Similar to the new species, most samples of L. atromarginatus (3/4), L. ferruginascens (3/4) and L. oomsisiensis (4/5) have ITS region with INDELs. These sequences were phased into two copies (clones A and B in Fig. 1), which were both included in the subsequent analysis. Only one of the ten specimens of the new species sequenced has an INDEL in the LSU region (GDGM 70573, GenBank accessions MW669931 and MW669932). ITS region of the new species has 12 informative sites among the 23 individuals and LSU has only two among the ten individuals. The ITS alignment had 726 characters: 275bp of ITS1 (complete), 154bp of 5.8S, and 297bp of ITS2 (complete).
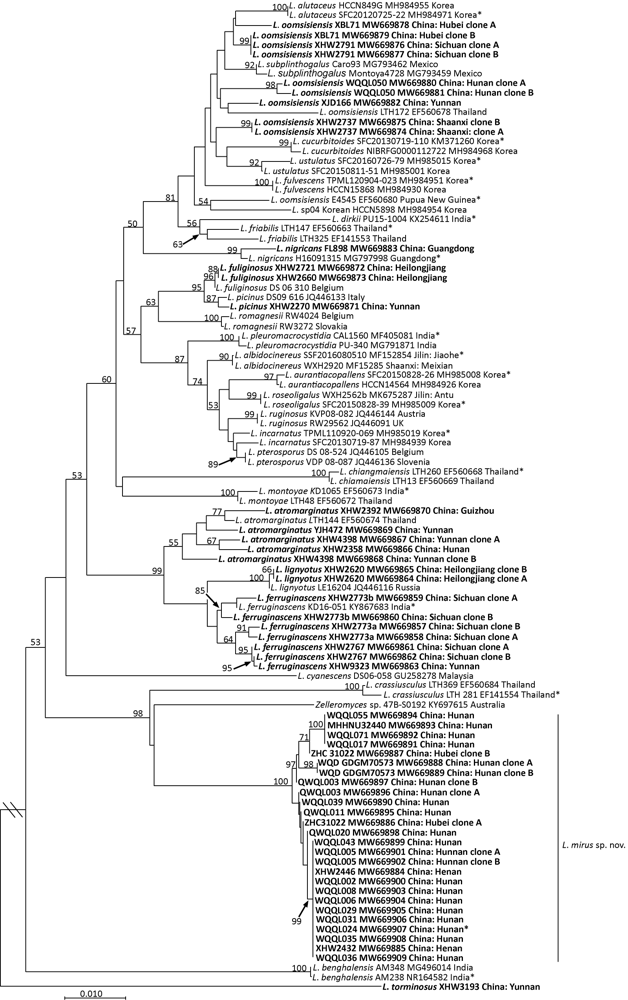
Fig. 1
Original tree generated by neighbor-joining (NJ) analysis of Lactarius mirus and its relatives using ITS sequence data, rooted with L. torminosus. NJ bootstrap proportions higher than 50% are indicated above the branches. Sample labels are provided in the order of species name, strain, GenBank accession and geographical origin. For Chinese samples, province names are given. Type specimens are marked with an asterisk. Sequences generated in this study are in bold.
2.2 Phylogenetic relationships
NJ analyses using ITS and LSU data produced bootstrap consensus trees with most of the backbones lacking supports (Figs. 1 and 2). Most high support values are on the branches leading to species or species complexes. In both trees, samples of the new species grouped into a terminal clade (both NJ-BP 100%) with relatively long branches. In the ITS tree, the new species, Thai L. crassiusculus and an Australian sample of Zelleromyces (KY697615) formed a highly supported clade (NJ-BP 98%), basal to the other clades (but only with NJ-BP 53%). In the ITS tree, the clades of L. atromarginatus and L. ferruginascens lacked significant supports and the samples with a morphology of L. oomsisiensis even did not form a monophyly.
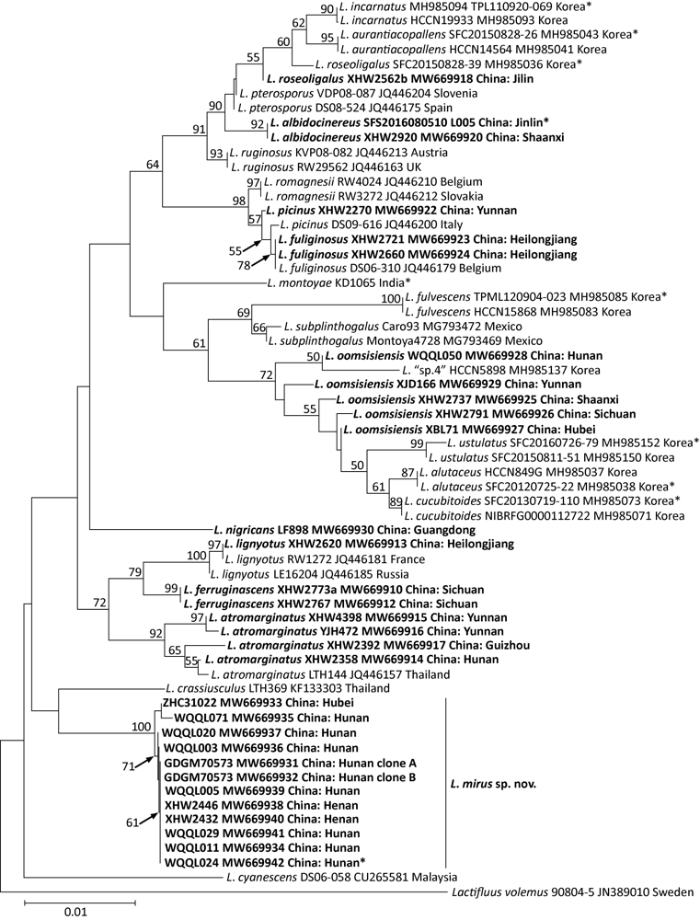
Fig. 2
Original tree generated by neighbor-joining (NJ) analysis of Lactarius mirus and its relatives using LSU sequence data, rooted with Lactifluus volemus. NJ bootstrap proportions higher than 50% are indicated above the branches. Sample labels are provided in the order of species name, strain, GenBank accession and geographical origin. For Chinese samples, province and county names are given. Type specimens are marked with an asterisk. Sequences generated in this study are in bold.
In the LSU tree, all the species received significant supports except that the samples of L. oomsisiensis did not group into a monophyly. The relationships among the new species and the other species in the ingroup were unsolved. It still formed a clade with L. crassiusculus, but this clustering lacked support.
2.3 Taxonomy
Lactarius mirus X.H. Wang, W.Q. Qin, Z.H Chen, W.Q. Deng & Zhen Wang, sp. nov.Fig. 3
Fig. 3
Basidiocarps and microscopic drawings of Lactarius mirus. A, C and F from holotype; B, D, E and G from KUN-HKAS112558. A, B: Basidiocarps; C: Basidiospores (holotype); D: Lamellar edge; E: Hymenophoral pseudocystidia; F: Pileipellis; G: Stipitipellis.
MycoBank MB838936
Etymology: referring to the beautiful extraordinary yellowing latex.
Diagnosis: Lactarius mirus is characterized by the grayish brown pileus, distant lamellae, brightly yellowing latex and spore ornamentation up to 3.0µm high. It differs from Malaysian L. pallidior by the much darker pileus and stipe and much higher spore ornamentation; from L. flavorosescens by the bigger spores with branching other than zebroid ridges.
Holotype: CHINA. Hunan Prov., Zhangjiajie, Yongding District, Zhangjiajie Campus of Jishou University, N29°08ʹ32″ E110°27ʹ37″, elev. 230m, in forest dominated with Pinus massoniana, Quercus serrata, Q. fabri and Q. aliena var. acutiserrata, 20 September 2020, W.Q. Qin, no. L024 (HKAS 110957, KUN!).
Habit and habitat: solitary or 2-3 individuals growing together, in fagaceous forests, from June to October.
Basidiocarps medium-sized, thick-fleshed. Pileus 40-80mm diam., convex to plano- convex with an involute margin when young, becoming applanate to wavy concave; surface smooth to faintly rugulose at the center and radially rugose or radially grooved in the marginal zone when mature, margin ± crenulate, dry to moist, subvelvety, grayish brown (6E2-6E3) with irregular paler mottles. Context 2-3mm thick, whitish, soon staining vivid yellow (3A7) by the latex, drying becoming apricot yellow or orange. Stipe 25-45×6-10mm, cylindrical, equal or tapering downwards, central to eccentrical; surface dry and subvelvety, concolorous with or paler than pileus. Lamellae sinuately attached to decurrent, distant, rather thick, 5mm broad, thick, whitish when young, warm cream- colored (4A3) when mature, instantly staining bright yellow by latex; the edge smooth and concolorous with the surface. Latex copious, white, instantly changing to vivid yellow (3A7), drying remaining so or further changing to apricot yellow to orange brown; taste acrid. Spore print ochraceous.
Basidiospores (7.0)8.0-8.9-10.0(11.5)× 7.0-8.5-9.5(11.0)µm [Q=1.00-1.13(1.17), Q=1.05±0.03] (440/22/18), subglobose to globose; ornamentation (1.5)2.0-3.0(3.5)µm high, composed of high, acute and longer ridges and transparent wings, some shorter and irregular ridges and warts present between the main ridges, ridges often branched, less amyloid in the upper part and often looking as if split, forming a reticulate pattern but rarely forming closed meshes, very rarely in a slightly spiral pattern, base of main ridges up to 1µm wide; plage mostly distally amyloid, rarely totally amyloid. Basidia 45-65(77)×13-17µm, 4-spored, subclavate, subfusiform, sterigmata 5-7µm long. Pleuromacrocystidia absent. Hymenophoral pseudocystidia 3-6µm diam., common to numerous, hyaline to pale golden brown, tortuous, often forking, buried in hymenium, rarely projecting. Lamellar edge sterile; cheilomacrocystidia absent; marginal cells 20-40×4-6µm, often bending, subventricose, sublageniform, hyaline, thin-walled. Hymenophoral trama with hyphoid cells and abundant golden brownish lactifers, lacking rosettes. Pileipellis a trichoepithelium, some parts a transition between a (ixo-) trichoepithelium to hymenoepithelium, 60-100µm thick, yellowish brown intracellular pigmentation present in the upper 2/3 part to the whole pellis; terminal cells of suprapellis 15-50(60)×4-7µm, cylindrical, ventricose, clavate, often gelatinized, i.e. hyphal walls not easily separated and outlines ambiguous; subpellis composed of inflated cells and hyphoid elements, cells 10-30(45)×10- 20(35)µm, slightly gelatinized. Stipitipellis a trichoderm or a transition between a cutis to trichoderm, 30-60µm thick; hyphae 3-6(8)µm in diameter, not gelatinized to gelatinized, yellowish brown; terminal cells 20-50×4-6µm, cylindrical, ventricose, subclavate or tapering. Trama of pileus and stipe with rosettes and abundant golden-brown lactifers. Clamp connections absent.
Additional specimens examined: CHINA. Chongqing: Wushan, N31°06ʹ46.86″ E110°04ʹ11.56″, elev. 1 185m, in forest dominated with Quercus aliena, 12 August 2019, J.H. Zhang, s.n. (HKAS112557). Henan: Neixiang Co., Xiaguan Town, Taohuayuan, in oak forest, 31 July 2010, N33°25ʹ42″ E111°49ʹ10″, elev. 460m, X.H. Wang, no. 2432 (KUN-HKAS61746); ibid., in oak forest, N33°26ʹ10″ E111°48ʹ58″, elev. 480m, 1 August 2010, X.H. Wang, no. 2446 (KUN-HKAS61754). Hubei: Enshi City, southeastern suburb, N30°16ʹ19″ E109°29ʹ17″, elev. 1 000m, 20 July 2017, Z.H. Chen, no. 31022 (KUN-HKAS112558). Hunan: Guiyang Co., Dongcheng Town, Xinwuchang, N25°58ʹ40″ E112°50ʹ23″, elev. 300m, 10 July 2017, W.Q. Deng, no. GDGM70573 (GDGM); Zhangjiajie, Yongding District, Tianmen Mt., N29° 03ʹ1.85″ E 110°28ʹ44.73″, elev. 1 471m, in forest dominated with Cyclobalanopsis multinervis, Fagus lucida and Q. engleriana, 20 July 2020, W.Q. Qin, no. L055 (KUN-HKAS111323); ibid., 1 August 2020, W.Q. Qin, no. L071 (KUN-HKAS111339); ibid., 3 September 2020, W.Q. Qin, no. L017 (KUN-HKAS110940); ibid., N29°03ʹ2.83″ E110°28ʹ37.39″, elev. 1 456m, Z.H. Chen, no. 32440 (MHHNU); Zhangjiajie, Yongding District, Zhangjiajie Campus of Jishou University, in forest dominated with Pinus massoniana, Q. serrata, Q. fabri and Q. aliena var. acutiserrata, 14 June 2020, W.Q. Qin, no. L002 (KUN-HKAS110920), no. L003 (KUN-HKAS110921); ibid., 16 June 2020, W.Q. Qin, no. L005 (KUN-HKAS110923), L006 (KUN-HKAS110924); ibid., 18 June 2020, W.Q. Qin, no. L008 (KUN-HKAS110926); ibid., 23 June 2020, W.Q. Qin, no. L011 (KUN-HKAS110929); ibid., 17 September 2020, W.Q. Qin, no. L020 (KUN-HKAS110943); ibid, 26 September 2020, W.Q. Qin, no. L029 (KUN-HKAS110953), no. L031 (KUN-HKAS110955); ibid., 28 September 2020, W.Q. Qin, no. L035 (KUN-HKAS110959), no. L039 (KUN-HKAS110960); ibid., 2 October 2020, W.Q. Qin, no. L039 (KUN-HKAS110963); ibid., 9 October 2020, W.Q. Qin, no. L043 (KUN-HKAS110967).
3 DISCUSSION AND CONCLUSION
Compared with those of Lactarius subg. Russularia and L. subg. Lactarius, species of L. subg. Plinthogalus are more homogenous in morphology. Pileus and stipe color at most vary from whitish gray to dark brown, never brightly colored as in the other two subgenera. The pileipellis is mostly a trichopalisade or hymenoepithelium and spores are subglobose to globose. Macrocystidia are only reported in very few species (Hesler & Smith 1979; Heilmann-Clausen et al. 1998; Le et al. 2007; Uniyal et al. 2019). Likewise, DNA sequence data are highly similar. ITS sequence similarities among different species often reach 98%, which are shown as very short branches in phylogenetic trees (e.g. Stubbe & Verbeken 2012; Shi et al. 2018; Lee et al. 2019). Species delimitation of L. subg. Plinthogalus now relies on the combination of barcode ITS region (often used with LSU) and subtle morphological differentiation, mostly pileus color, discoloration of latex and spore ornamentation and less powerful the length of terminal cells of pileipellis. These are the criteria that we used here to determine the identity of L. mirus.
The species described by Stubbe et al. ( 2007, 2008) from Malaysia updated our understanding on the morphological diversity of this group. Species with bluing latex or context (Lactarius cyanescens, L. lazulinus and L. mirabilis) were reported for the first time in the subgenus. In L. lazulinus and L. mirabilis between the initial white and the final bluish staining, there is a yellow change. Later two additional species, L. pallidior and L. flavorosescens, were described from Malaysia, both having a pinkish color change after the yellow change. Le et al. (2007) noticed one specimen of Thai L. crassiusculus, the closest relative of L. mirus shown in our phylogenetic trees, has a yellowish discoloration on the context after turning pink. In L. mirus, the latex remained yellow or changed to deep apricot yellow or orange-brown when dry, never with the bluish or pinkish discoloration observed on the Southeast Asian species. The bright yellow color is undoubtedly much stronger and remarkable than the “yellow-ocher” discoloration of North American L. subvernalis.
None of the species mentioned above have spore ornamentation as high as Lactarius mirus, which are mostly 2.0-3.0µm with extremity to 3.5µm. In the field L. mirus resembles L. flavorosescens. They have basidiocarps with firm stature, pileus and relatively short stipe both in a chocolate brown color, and broad, distant and dark cream lamellae in common. The spore ornamentation of L. flavorosescens is typically zebroid, lower (<2.5µm) and rarely branching. The spore ornamentation of L. pallidior is similar to that L. mirus, but much lower [1.5-2(2.5)µm high]. Moreover, L. pallidior has a nearly white pileus. The Thai L. crassiusculus has abundant hymenophoral macrocystidia and close lamellae, therefore it is impossible to confuse it with L. mirus. Considering the accepted species of L. subg. Plinthogalus often have only one or two morphological differences, we conclude it is sufficient to describe a new species based on these specimens.
The finding of species with a yellow color change in China adds evidence showing that species with milky caps in subtropical-tropical China have strong affinity with those in the tropics, especially Southeast Asia. Based on morphology, we suppose that the closest relatives of Lactarius mirus are most likely Malaysian L. pallidior and L. flavorosescens. In this paper, besides the DNA data of the new species, we provided ITS and LSU sequences from Chinese samples of Lactarius oomsisiensis and L. atromarginatus. Both species are described from Pupua New Guinea and later found from Thailand. Although in the ITS tree, the support value for L. atromarginatus is not significant, the Chinese samples grouped with a Thai sample (LTH144) and morphologically they are not separable. With the inclusion of ITS and LSU sequences from more Chinese “L. oomsisiensis” samples into the analysis, the species limit of L. oomsisiensis becomes ambiguous and needs to be re-assessed. Nevertheless, the Chinese samples are still closer to Thai sample and the holotype from Papua New Guinea. In L. subg. Lactarius and L. subg. Russularia, subtropical China shares quite many species with Thailand, Vietnam and even Papua New Guinea, such as L. ambiguus X.H. Wang, L. inconspicuous H.T. Le & F. Hampe, L. liliputianus, L. rubrobrunneus H.T. Le & Nuytinck, L. laccarioides Wisitr. & Verbeken, L. austrozonarius H.T. Le & Verbeken and L. sulphurescens Verbken & E. Horak (Wang 2018, 2019; Chinese specimens identified by X.H. Wang). In the other genera of Russulaceae, such connection is also observed (Wang et al. 2015, 2018b; Rossi et al. 2020). Ecologically most of these species are found in subtropical-tropical forests dominated by fagaceous trees. The similar phenomenon was found in wood-inhabiting fungi (Chen et al. 2016; Wu et al. 2020). We believe the growing taxonomic study of Russulaceae in subtropical-tropical China, especially using specimens collected from broad-leaved forests, will eventually establish a link between the subtropical and tropical mycofloras of Asia.
Acknowledgments:
We thank S.Q. Cao, Kunming Institute of Botany, Chinese Academy of Sciences for doing lab work and microscopical measurements. The specimen of L. nigricans is provided by Dr. F. Li, Sun Yat-Sen University, China
参考文献
Global diversity and molecular systematics of Wrightoporia s.l. (Russulales, Basidiomycota)
DOI:10.3767/003158516X689666
PMID:28232759
[本文引用: 1]

Wrightoporia accommodates polypores producing finely asperulate and amyloid basidiospores, and causing white rot. Thirty-nine species have been described or transferred to this genus; however, only a few species have been referred to molecular phylogeny. In this study, about 140 worldwide specimens of Wrightoporia s.l. were studied morphologically, and ITS and/or nLSU regions from 37 samples, representing 19 species, were sequenced for phylogenetic analysis. Six clades of Wrightoporia s.l. were recognized. The Wrightoporia s. str. clade includes W. avellanea, W. lenta (the generic type) and W. subavellanea. Three clades segregating from Wrightoporia s. str. were proposed separately as three new genera, namely Larssoniporia gen. nov., Pseudowrightoporia gen. nov. and Wrightoporiopsis gen. nov. Two other clades were named after Amylonotus and Amylosporus. According to phylogenetic and morphological evidence, species previously treated in Wrightoporia were transferred to Amylonotus, Amylosporus and the new genera, or were retained as members of Wrightoporia s.l. because no good solution for these species could be found so far. In addition, one new species in Larssoniporia, three new species in Pseudowrightoporia and two new species in Wrightoporiopsis were described. Identification keys to the six genera and species in Amylonotus, Amylosporus, Larssoniporia, Pseudowrightoporia, Wrightoporia and Wrightoporiopsis are provided, respectively.
Morphological and phylogenetic evidence for two new Lactarius species (Russulales, Basidiomycota) from India
DOI:10.7872/crym/v38.iss4.2017.453 URL [本文引用: 1]
BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT
The genus Lactarius: fungi of northern Europe. Vol. 2
Evolutionary consequences of putative intra-and interspecific hybridization in agaric fungi
DOI:10.3852/13-041
PMID:23928423
[本文引用: 1]

Agaric fungi of the southern Appalachian Mountains including Great Smoky Mountains National Park are often heterozygous for the rDNA internal transcribed spacer region (ITS) with >42% of collections showing some heterozygosity for indels and/or base-pair substitutions. For these collections, intra-individual haplotype divergence is typically less than 2%, but for 3% of these collections intra-individual haplotype divergence exceeds that figure. We hypothesize that high intra-individual haplotype divergence is due to hybridization between agaric fungi with divergent haplotypes, possibly migrants from geographically isolated glacial refugia. Four species with relatively high haplotype divergence were examined: Armillaria mellea, Amanita citrina f. lavendula, Gymnopus dichrous and the Hygrocybe flavescens/chlorophana complex. The ITS region was sequenced, haplotypes of heterozygotes were resolved through cloning, and phylogenetic analyses were used to determine the outcome of hybridization events. Within Armillaria mellea and Amanita citrina f. lavendula, we found evidence of interbreeding and recombination. Within G. dichrous and H. flavescens/chlorophana, hybrids were identified but there was no evidence for F2 or higher progeny in natural populations suggesting that the hybrid fruitbodies might be an evolutionary dead end and that the genetically divergent Mendelian populations from which they were derived are, in fact, different species. The association between ITS haplotype divergence of less than 5% (Armillaria mellea = 2.6% excluding gaps; Amanita citrina f. lavendula = 3.3%) with the presence of putative recombinants and greater than 5% (Gymnopus dichrous = 5.7%; Hygrocybe flavescens/chlorophana = 14.1%) with apparent failure of F1 hybrids to produce F2 or higher progeny in populations may suggest a correlation between genetic distance and reproductive isolation.
Fungal diversity notes 253-366: taxonomic and phylogenetic contributions to fungal taxa
DOI:10.1007/s13225-016-0366-9 URL [本文引用: 1]
Recent developments in the MAFFT multiple sequence alignment program
DOI:10.1093/bib/bbn013 URL [本文引用: 1]
MEGA X: Molecular evolutionary genetics analysis across computing platforms
DOI:10.1093/molbev/msy096 URL [本文引用: 1]
Lactarius in Northern Thailand: 2. Lactarius subgenus Plinthogali
Lactarius cucurbitoides (Russulales, Basidiomycota), a new species from South Korea supported by molecular and morphological data
DOI:10.11646/phytotaxa.205.3 URL [本文引用: 1]
Taxonomic revision of the genus Lactarius (Russulales, Basidiomycota) in Korea
DOI:10.1007/s13225-019-00425-6 URL [本文引用: 8]
Phylogenetic relationships of agaric fungi based on nuclear large subunit ribosomal DNA sequences
Phylogenetic relationships of mushrooms and their relatives within the order Agaricales were addressed by using nuclear large subunit ribosomal DNA sequences. Approximately 900 bases of the 5' end of the nucleus-encoded large subunit RNA gene were sequenced for 154 selected taxa representing most families within the Agaricales. Several phylogenetic methods were used, including weighted and equally weighted parsimony (MP), maximum likelihood (ML), and distance methods (NJ). The starting tree for branch swapping in the ML analyses was the tree with the highest ML score among previously produced MP and NJ trees. A high degree of consensus was observed between phylogenetic estimates obtained through MP and ML. NJ trees differed according to the distance model that was used; however, all NJ trees still supported most of the same terminal groupings as the MP and ML trees did. NJ trees were always significantly suboptimal when evaluated against the best MP and ML trees, by both parsimony and likelihood tests. Our analyses suggest that weighted MP and ML provide the best estimates of Agaricales phylogeny. Similar support was observed between bootstrapping and jackknifing methods for evaluation of tree robustness. Phylogenetic analyses revealed many groups of agaricoid fungi that are supported by moderate to high bootstrap or jackknife values or are consistent with morphology-based classification schemes. Analyses also support separate placement of the boletes and russules, which are basal to the main core group of gilled mushrooms (the Agaricineae of Singer). Examples of monophyletic groups include the families Amanitaceae, Coprinaceae (excluding Coprinus comatus and subfamily Panaeolideae), Agaricaceae (excluding the Cystodermateae), and Strophariaceae pro parte (Stropharia, Pholiota, and Hypholoma); the mycorrhizal species of Tricholoma (including Leucopaxillus, also mycorrhizal); Mycena and Resinomycena; Termitomyces, Podabrella, and Lyophyllum; and Pleurotus with Hohenbuehelia. Several groups revealed by these data to be nonmonophyletic include the families Tricholomataceae, Cortinariaceae, and Hygrophoraceae and the genera Clitocybe, Omphalina, and Marasmius. This study provides a framework for future systematics studies in the Agaricales and suggestions for analyzing large molecular data sets.
Fungal biodiversity profiles 91-100
DOI:10.5252/cryptogamie-mycologie2020v41a4 URL [本文引用: 1]
Three new species of Lactarius (Russulaceae, Russulales) from Northeast China
DOI:10.1016/j.myc.2017.11.001 URL [本文引用: 3]
Lactarius subgenus Plinthogalus of Malaysia
Critical assessment of the Lactarius gerardii species complex (Russulales)
DOI:10.1016/j.funbio.2010.01.008
PMID:20943137
[本文引用: 1]

This paper investigates species delimitation within the Lactarius gerardii species complex and explores its taxonomic and geographical extent. A combined molecular phylogeny based on ITS, LSU and rpb2 gene sequences is constructed and morphological characters are evaluated. While L. gerardii was originally described from North America, it has later been reported from all over Asia. Therefore a worldwide sampling range was aimed at, including species exhibiting morphological affinities with L. gerardii. The phylogenetic analyses indicate that intercontinental conspecificity in L. gerardii is absent. Thirty strongly supported clades are retrieved of which 18 are morphologically identifiable species. The group is elevated to Lactarius subg. Gerardii stat. nov. It includes, apart from L. gerardii s.l., L. atrovelutinus, L. bicolor, L. ochrogalactus, L. petersenii, L. reticulatovenosus, L. sepiaceus, L. subgerardii and L. wirrabara, as well as the pleurotoid L. uyedae. The paraphyletic nature of the genus Lactarius is confirmed. Lactarius subg. Gerardii appears not affiliated with L. subg. Plinthogalus and this can be substantiated morphologically. No representatives are known from Europe, Africa or South America. The high frequency of intercontinental sister relationships observed between America, Asia and the Australian region, suggests multiple migration and speciation events have occurred across continents.Copyright © 2010 The British Mycological Society. Published by Elsevier Ltd. All rights reserved.
Blue-staining species of Lactarius subgenus Plinthogali in Malaysia
Lactarius subg. Plinthogalus: the European taxa and American varieties of L. lignyotus re-evaluated
DOI:10.3852/12-039
PMID:22684287
[本文引用: 4]

The European species Lactarius subg. Plinthogalus were subjected to a molecular phylogenetic analysis based on ITS, LSU and rpb2 sequences. Morphological characters of the species are discussed in the light of the phylogenetic results. In addition to a broad sampling within Europe, some Asian and North American taxa also were included in the analysis. Eight European species are confirmed molecularly: L. lignyotus, L. acris, L. azonites, L. pterosporus, L. ruginosus, L. romagnesii, L. fuliginosus and L. picinus. Except the sibling species L. fuliginosus and L. picinus, all are morphologically distinct. Our results suggest that L. fuliginosus is associated exclusively with broadleaf trees and L. picinus with conifers, but this putative difference in host specificity needs to be investigated further. Lactarius subruginosus turns out to be a synonym of either L. pterosporus or L. ruginosus. The position of Lactarius terenopus remains to be clarified. The North American taxa that are closely related to the European L. lignyotus (L. fallax, L. lignyotus var. canadensis, var. nigroviolascens, var. marginatus) are not resolved. Intercontinental conspecificity was demonstrated between Europe and northern Asia but was not found between Europe and southern Asia or between Europe and North America. A taxonomic subdivision of L. subg. Plinthogalus based on the height of the spore ornamentation should be rejected.
New combinations in Lactifluus. 2. L. subgenus Gerardii
DOI:10.5248/119.483 URL [本文引用: 3]
Lactarius pleuromacrocystidiatus (Russulaceae), a novel species from India
DOI:10.1007/s12225-018-9788-8 URL [本文引用: 2]
Lactarius (Basidiomycetes) in Papua New Guinea 2. Species of tropical montane rainforests
DOI:10.1071/SB99023 URL [本文引用: 1]
Tales of the unexpected: angiocarpous representatives of the Russulaceae in tropical South East Asia
DOI:10.3767/003158514X679119
PMID:25264381
[本文引用: 2]

new sequestrate Lactarius species are described from tropical forests in South East Asia. Extensive macro- and microscopical descriptions and illustrations of the main anatomical features are provided. Similarities with other sequestrate Russulales and their phylogenetic relationships are discussed. The placement of the species within Lactarius and its subgenera is confirmed by a molecular phylogeny based on ITS, LSU and rpb2 markers. A species key of the new taxa, including five other known angiocarpous species from South East Asia reported to exude milk, is given. The diversity of angiocarpous fungi in tropical areas is considered underestimated and driving evolutionary forces towards gasteromycetization are probably more diverse than generally assumed. The discovery of a large diversity of angiocarpous milkcaps on a rather local tropical scale was unexpected, and especially the fact that in Sri Lanka more angiocarpous than agaricoid Lactarius species are known now.
Lactarius verrucosporus sp. nov. and L. nigricans sp. nov., two new species of Lactarius (Russulaceae) from southern China
DOI:10.11646/phytotaxa.364.3 URL [本文引用: 1]
Fungal biodiversity profiles 71-80
DOI:10.7872/crym/v39.iss4.2018.419 URL [本文引用: 1]
Two new species of Lactarius from Changbai and Lesser Khingan Mountains
Revisiting the morphology and phylogeny of Lactifluus with three new lineages from southern China
DOI:10.3852/13-393 URL [本文引用: 1]
Phylogeny, biogeography and taxonomic reassessment of Multifurca (Russulaceae, Russulales) using three-locus data
DOI:10.1371/journal.pone.0205840 URL [本文引用: 1]
Lactifluus parvigerardii sp. nov., a new link towards the pleurotoid habit in Lactifluus subgen. Gerardii (Russulaceae, Russulales)
DOI:10.7872/crym.v33.iss2.2012.181 URL [本文引用: 1]
Lactarius subgenus Russularia (Basidiomycota, Russulales): novel Asian species, worldwide phylogeny and evolutionary relationships
DOI:S1878-6146(16)30106-4
PMID:27890091
[本文引用: 1]

Lactarius subg. Russularia is a large group of milkcaps occurring almost worldwide and dominant in many ecosystems. In this study we focus on new diversity, evolutionary relationships, divergence time, and origin of the subgenus. Six conifer symbionts are described as new to science: Lactarius atrii, L. aurantionitidus, L. dombangensis, L. flavigalactus, L. lachungensis, and L. sikkimensis. Species delimitation is assessed based on the concordance between morphological characteristics and an ITS phylogeny. Infrageneric relationships were studied using a phylogeny constructed from concatenated ITS-rpb2 data using Maximum Likelihood and Bayesian inference. Results show that species in this subgenus do not cluster together according to their geographic origin. Intercontinental sister relationships between Europe/Asia/North America are common but actual conspecificity is rare. This result suggests that allopatric speciation has played an important role within this subgenus. Only few morphological characteristics tend to be phylogenetically informative, with the most important being presence or absence of true cystidia and the pileipellis structure. Two datasets were generated in order to estimate the age of L. subg. Russularia. The results suggest the origin of L. subg. Russularia to be in the Mid Miocene period.Copyright © 2016 British Mycological Society. Published by Elsevier Ltd. All rights reserved.
Polypore diversity in South China