Diaporthe novem属子囊菌门Ascomycota,粪壳菌纲Sordariomycetes,间座壳目Diaporthales,间座壳科Diaporthaceae,间座壳属Diaporthe,其无性阶段为Phomopsis sp.9 (van Rensburg et al. 2006)。它与严重危害大豆的大豆北方茎溃疡病菌D. caulivora、大豆南方茎溃疡病菌D. aspalathi和大豆拟茎点种腐病菌D. longicolla互为近似种,其中大豆北方茎溃疡病菌和大豆南方茎溃疡病菌被列入我国进境植物检疫性有害生物名录。
D. novem最早由Santos et al. (2011)正式命名,现有资料显示该病菌在智利(Díaz et al. 2014)、克罗地亚(Santos et al. 2011)、意大利(Rekab et al. 2004;Dissanayake et al. 2017;Guarnaccia & Crous 2017)、葡萄牙(Santos et al. 2010)、南非(van Niekerk et al. 2005;van Rensburg et al. 2006)和澳大利亚(Thompson et al. 2018)等国家有分布。危害大豆(Santos et al. 2011;Petrović et al. 2018)、向日葵(Rekab et al. 2004;Thompson et al. 2018)、猕猴桃(Díaz et al. 2014;Díaz et al. 2017)、葡萄(van Niekerk et al. 2005)、金橘和青柠(Guarnaccia & Crous 2017)等重要经济作物,引起种子和果实腐烂,造成严重经济损失。该菌也在绣球花(Santos et al. 2010)、南非红茶(van Rensburg et al. 2006)、茜草科拉拉藤属(Dissanayake et al. 2017)上有分离报道,我国尚未见报道。
2019年8月,天津海关动植物与食品检测中心植物检疫实验室从进境美国大豆中分离培养获得了1株疑似间座壳属菌株MDD57,对其开展了形态研究,并进行了测序和致病性测定,确定该病菌为D. novem,现将结果报道如下。
1 材料与方法
1.1 材料
供试样品为2019年8月到岸的美国进境大豆种子。致病性测定所用植株为栽培3周的健康黄大豆幼苗。
1.2 方法
1.2.1 病原菌的分离培养
挑取皱缩、带有病斑的病豆,用1%次氯酸钠表面消毒5 min,灭菌水冲洗3次,置25 ℃下保湿培养24 h,-20 ℃冰冻24 h,置PDA培养基上,22 ℃ 12 h黑暗/光照交替培养。5 d后观察培养结果,对可疑菌落用PDA纯化。对纯化菌落镜检,记录菌落特征,测定分生孢子形态及大小。
1.2.2 DNA提取、PCR扩增和测序
经纯化的可疑菌落,培养7 d左右,取适量菌丝块,液氮充分研磨后,用植物基因提取试剂盒(德国Qiagen公司Dneasy plant mini kit)提取DNA。PCR反应采用真菌通用引物ITS4和ITS5进行ITS片段扩增(White et al. 1990),采用引物EF1-728F和EF1-986R进行tef1α片段扩增(Carbone & Kohn 1999),2对引物由生工生物工程(上海)股份有限公司合成。具体引物序列及反应条件见表1。PCR反应体系总体积25 μL,包含:EmeraldAmp MAX PCR Master Mix (2× Premix) 12.5 μL;ddH2O 9.0 μL;引物各1.0 μL;DNA模板1.5 μL (30 ng/μL)。PCR相关试剂由TaKaRa公司生产。扩增产物经2.0%琼脂糖凝胶在1×TAE缓冲液中电泳,确认扩增成功后,产物送生工生物工程(上海)股份有限公司北京测序部测序。
表1 引物序列及反应条件
Table 1
| 序号 No. | 引物名称 Primer | 引物序列 Primer sequences (5°→3°) | 反应条件 PCR conditions |
|---|---|---|---|
| 1 | ITS4 | TCCTCCGCTTATTGATATGC | 94 °C 3 min; (94 °C 30 s, 58 °C 30 s, 72 °C 30 s) 35 cycles; 72 °C 5 min |
| ITS5 | GGAAGTAAAAGTCGTAACAAGG | ||
| 2 | EF1-728F | CATCGAGAAGTTCGAGAAGG | 94 °C 5 min; (94 °C 45 s, 52 °C 30 s, 72 °C 90 s) 40 cycles; 72 °C 6 min |
| EF1-986R | TACTTGAAGGAACCCTTACC |
1.2.3 序列比对和系统发育分析
利用DNAMAN 8.0分析软件对测得的序列进行装配;序列同源性比较分析采用BLAST (Basic Local Alignment Search Tools)工具(
1.2.4 致病性测定
将分离纯化后的病原菌转至PDA培养基,于22 ℃ 12 h黑暗/光照交替培养10 d。挑取菌落中适量的分生孢子液,用无菌水制成分生孢子悬浮液,孢子适宜浓度约为1×107 个/mL。
选用栽培3周的健康黄大豆幼苗,用70%酒精对幼苗叶片及茎基部进行擦拭消毒。叶片采用针刺法接种分生孢子悬浮液,茎基部采用创伤法接种菌丝。用湿润的灭菌脱脂棉覆盖伤口,保湿1 d,置于20-22 ℃下生长观察。同时设置无菌水空白对照。出现症状后,对发病叶片及茎基部的病健交界组织进行病原菌再分离。
2 结果与分析
2.1 病菌的培养性状和形态学特征
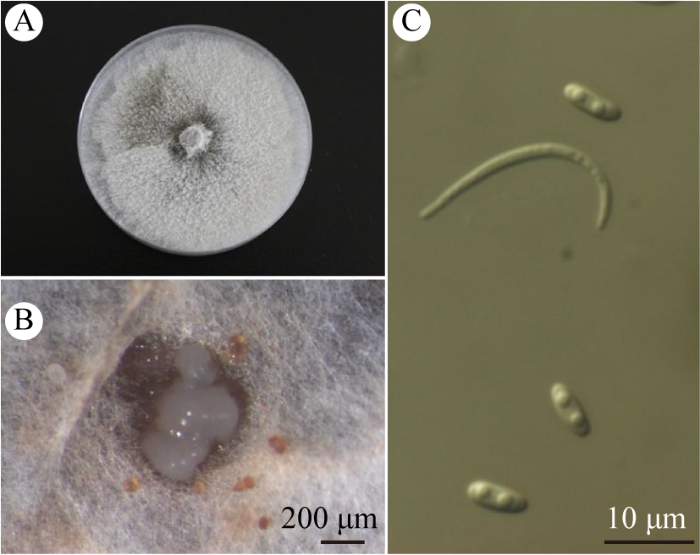
菌株MDD57在PDA培养基上,菌落培养初期白色,菌丝絮状、致密,边缘不整齐。菌落生长迅速,培养1周左右即可长满培养皿,此时菌落中间呈浅棕色或橄榄色,随着培养时间的增加,颜色逐渐加深并扩散至菌落部分区域,呈深橄榄色或灰黑色(图1A)。培养2周左右,菌落表面可见零星分生孢子器,随着分生孢子器的成熟,有时可见乳白色分生孢子液溢出(图1B)。分生孢子单胞、无色、透明。α型孢子纺锤形,两端稍钝,无隔膜,通常含2个油球,大小为 6.0-10.0×2.0-3.0 μm,平均7.0×2.3 μm。β型孢子为长针状,一端常钩状弯曲,大小为25.0-36.0× 1.2-2.0 μm,平均29.6×1.4 μm (图1C)。
图1
图1
MDD57在PDA上的菌落及其形态特征
A:PDA培养7 d后的菌落;B:分生孢子液;C:α型分生孢子和β型分生孢子
Fig. 1
Colony and morphological characteristics of strain MDD57 on PDA.
A: Colony on PDA (7 d); B: Conidial drop; C: α-conidia and β-conidia.
2.2 ITS和tef1α序列比对结果
将MDD57的ITS基因序列与NCBI网站GenBank中相关序列进行BLAST比对。结果显示,其与D. novem (登录号:MH299960)和D. helianthi (登录号:AJ312366)的相似度均为99%,通过ITS基因能确定MDD57为Diaporthe/ Phomopsis属真菌。
菌株MDD57的tef1α基因序列BLAST结果显示,其与D. novem (登录号:MH025961、MH025962)相似度均为100%。
2.3 系统发育分析
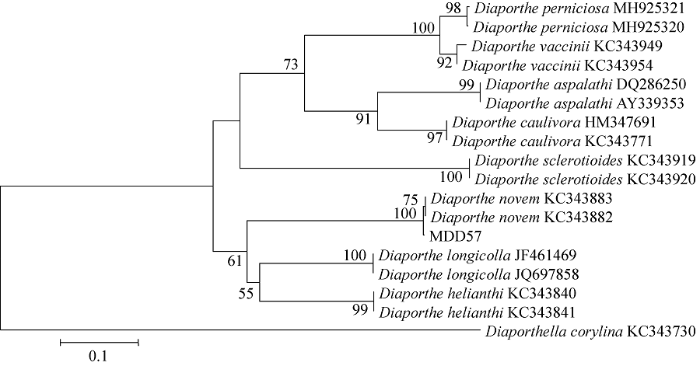
基于tef1α基因序列构建的系统发育分析结果见图2,所列几个种均可形成独立分支,MDD57与D. novem聚为一支。
图2
图2
基于间座壳属真菌MDD57及相关种类的tef1α序列采用邻接法构建的系统发育树
Fig. 2
Phylogenetic tree generated from tef1α sequences of Diaporthe novem strain MDD57 and related species based on neighbor-joining method (NJ).
2.4 致病性测定结果
MDD57接种大豆5 d左右开始发病。叶片接种部位出现浅褐色病斑,造成叶片皱缩、病斑周围变黄;大豆幼苗茎基部接种部位变黑,出现长条状、棕褐色病斑(图3)。接种15 d后,整株枯萎,全部幼苗死亡。根据柯赫氏法则,从茎基部发病部位再次分离病菌,获得与MDD57形态和tef1α基因序列一致的菌株。
图3
图3
菌株MDD57接种大豆幼苗7 d后的症状
A,B:接种叶片皱缩病斑及对照;C,D:接种茎基部棕褐色长条形病斑及对照
Fig. 3
Artificial inoculation symptoms caused by Diaporthe novem strain MDD57 on soybean seedling in 7 days after inoculation.
A, B: Crinkle spot of inoculated leaves and uninfected leaves of experimental control; C, D: Brown streak of inoculated stem-base and the uninfected control.
3 结论与讨论
通过对美国大豆种子中可疑菌株进行分离纯化、形态学观察、分子检测及致病性测定,最终确定该菌株为Diaporthe novem。间座壳属真菌以植物病原菌和内生菌的形式存在于健康的叶片、茎、种子和根中,或以腐生菌的形式存在于各种寄主的腐烂组织中(Murali et al. 2006;Garcia-Reyne et al. 2011;Udayanga et al. 2011)。由于分布广泛、寄主多样、病原菌危害严重,该属中的一些种类是重要的检疫性有害生物。在《中华人民共和国进境植物检疫性有害生物名录》中有5种检疫性真菌病害与该属相关,分别是大豆北方茎溃疡病菌D. caulivora、大豆南方茎溃疡病菌D. aspalathi、向日葵茎溃疡病菌D. helianthi、苹果果腐病菌D. perniciosa和蓝莓果腐病菌D. vaccinii。因此,对间座壳属不同种类真菌进行准确地检疫鉴定非常重要。
表2 已发表的大豆上4种间座壳属近似种的形态比较
Table 2
| 阶段 Phage | Diaporthe novema,b (syn. D. pseudolongicolla) | Diaporthe longicollab,c,d | Diaporthe caulivorac,d (syn. D. phaseolorum var. caulivora) | Diaporthe aspalathic,d (syn. D. phaseolorum var. meridionalis) |
|---|---|---|---|---|
| 现用名 Current name | Diaporthe novem | Phomopsis longicolla | Phomopsis phaseoli | Phomopsis phaseoli |
| 菌落形态 (PDA) Cultural characteristics on PDA | 白色致密的气生菌丝 Fresh mycelia white, compact, aerial | 白色,呈密集的丛卷毛状, 有时部分区域显黄绿色 White, compact aerial mycelium with typical yellowish-green ring around the centre of colony | 丛毛状的白色菌落, 后期变为黄色至茶色 White colonies tufted and turn yellow to tan in later stage | 白色,丛生长毛, 老熟后变成深褐色 White, tufted long hair, dark brown after maturity |
| 无性型形态 Anamorph characteristics | α型分生孢子单胞,卵圆形或椭球形, 两端稍钝,透明,通常含2个油球, 大小为6.0-10.0×1.9-3.1 μm;β型分 生孢子透明,单胞,丝状,一端弯曲, 大小为19.5-37.7×0.9-2.0 μm Alpha conidia unicellular, oval or ellipsoidal, with obtuse ends, hyaline, 2-guttules, 6.0-10.0× 1.9-3.1 μm; beta conidia hyaline, unicellular, filiform, curved at one end, 19.5-37.7×0.9-2.0 μm | α型分生孢子透明,单胞, 梭形,含2个油球,大小 4.9-9.0×1.4-3.5 μm;β型 分生孢子极少见 Alpha conidia hyaline, unicellular, ellipsoidal, 2-guttules, 4.9-9.0× 1.4-3.5 μm; beta conidia rarely seen | 很少见 Rarely seen in culture | 很少见 Rarely seen in culture |
| 生殖方式 Sexuality | 异宗配合 Heterothallic | 无性生殖 Asexual | 同宗配合 Homothallic | 同宗配合 Homothallic |
| 有性型形态 Teleomorph characteristics | 子囊38.5-43.0×6.5-10.5 μm,棍棒状, 顶端可见折光环;子囊孢子8.6-13.1× 2.1-3.6 μm,透明,光滑,圆柱形, 中间有隔膜,有时分隔处有缢缩, 常含4个油球,有时无油球 Asci 38.5-43.0×6.5-10.5 μm, clavate, with visible refractive apical ring; ascospores 8.6-13.1×2.1-3.6 μm, hyaline, smooth, cylindrical, medianly septate, sometimes constricted at the septum, normally 4-guttules, sometimes eguttulate | 未发现 Not found | 子囊29.8-40.0×4.0-7.0 μm, 长棍棒状,顶部有清晰的折 光环;子囊孢子8.0-12.0× 2.4-4.0 μm,透明,纺锤形, 双胞,常含油球 Asci 29.8-40.0×4.0-7.0 μm, clavate, with conspicuous refractive apical ring; ascospores 8.0-12.0×2.4-4.0 μm, hyaline, fusoid, two-celled, normally guttules | 子囊34.8-41.1×4.8-8.7 μm, 长棍棒状,顶部有清晰的折 光环;子囊孢子7.4-13.2× 2.3-4.2 μm,无色,纺锤形, 双胞,含4个油球 Asci 34.8-41.1×4.8-8.7 μm, clavate, with conspicuous refractive apical ring; ascospores 7.4-13.2×2.3-4.2 μm, colourless, fusoid, two-celled, 4-guttules |
Note: aSantos et al.
间座壳属真菌形态特征有限,所以利用形态特征和培养性状难以对该属进行准确鉴定(段维军等 2016)。本研究将来自美国的大豆样品中分离得到的间座壳属菌株MDD57确定为D. novem,分子上主要依据tef1α基因序列分析。在试验中,ITS基因并没有将D. novem与其近似种准确区分开来。对于待测菌株,我们可以先考虑用ITS基因确定到属,再寻找合适基因进一步鉴定到种。
关于D. novem的命名,van Rensburg et al. (2006)从顶梢枯死的南非红茶树中分离到1株Phomopsis,由于当时没有观察到它的生殖方式,无法将其命名,延续van Niekerk et al. (2005)的编号系统,将其标记为Phomopsis sp.9。Novem是拉丁文数字9,Santos et al. (2011)从克罗地亚大豆中分离到该病菌,并提出了Diaporthe novem J.M. Santos, Vrandečić & A.J.L. Phillips的正式命名。Petrović et al. (2018)从塞尔维亚大豆种子中检出该病菌,由于形态特征与危害症状与D. longicolla相近,将该病害称为D. pseudolongicolla,并作为一种塞尔维亚大豆上的新发病害报道。
大豆作为重要的粮油兼用作物、饲料原料,在国内有着巨大的需求,是我国最重要的进口农产品之一,2019年我国大豆进口总量达到8 851万t (胡佳续等 2021)。我国口岸多次检出D. novem,由于该病菌不在检疫性名录内,其危害往往容易被忽视。该病菌引起大豆茎秆溃疡和种子腐烂(Petrović et al. 2018),导致大豆产量和品质下降。由受害种子生产的豆油及大豆榨油后所得饲料、肥料及豆饼等其他产品质量低下,不适合作为饲料使用。此外,该病菌寄主广泛,一旦随进境大豆传入我国,将会对我国大豆及其他农作物生产、农业生态造成难以估量的损失。因此,口岸在对进境大豆检疫鉴定时,有必要加强对该菌的关注。
致谢
感谢沈阳农业大学植物保护学院周如军教授在拉丁名方面的指导。
参考文献
Soybean stem canker: an emerging disease problem
A method for designing primer sets for the speciation studies in filamentous ascomycetes
DOI:10.2307/3761358 URL [本文引用: 1]
First report of Diaporthe novem causing postharvest rot of kiwifruit during controlled atmosphere storage in Chile
Identification and characterization of Diaporthe ambigua, D. australafricana, D. novem, and D. rudis causing a postharvest fruit rot in kiwifruit
DOI:10.1094/PDIS-10-16-1535-RE URL [本文引用: 1]
Molecular phylogenetic analysis reveals seven new Diaporthe species from Italy
DOI:10.5943/mycosphere URL [本文引用: 2]
Identification of the quarantine fungus Diaporthe helianthi from the corn seeds imported from Ukraine
Cutaneous infection by Phomopsis longicolla in a renal transplant recipient from Guinea: first report of human infection by this fungus
Emerging citrus diseases in Europe caused by species of Diaporthe
DOI:10.5598/imafungus.2017.08.02.07
PMID:29242778
[本文引用: 2]

Species of are considered important plant pathogens, saprobes, and endophytes on a wide range of plant hosts. Several species are well-known on citrus, either as agents of pre- or post-harvest infections, such as dieback, melanose and stem-end rot on fruit. In this study we explored the occurrence, diversity and pathogenicity of species associated with and allied genera in European orchards, nurseries, and gardens. Surveys were carried out during 2015 and 2016 in Greece, Italy, Malta, Portugal, and Spain. A total of 79 strains were isolated from symptomatic twigs, branches and trunks. A multi-locus phylogeny was established based on five genomic loci (ITS,,, and ), and the morphological characters of the isolates determined. Preliminary pathogenicity tests were performed on lemon, lime, and orange plants with representative isolates. The most commonly isolated species were. and., while only four isolates of. were collected. Two new species, described here as. and. spp. nov. were found associated with a new devastating dieback disease of lemon plants. Furthermore, one cluster of sterile isolates was renamed as.. Pathogenicity tests revealed most of the species as susceptible to.,. and.. Moreover,. and. caused serious cankers affecting all the species tested. This study is the first report of and. on citrus in Europe, and the first detection of a new canker disease of citrus in Europe. However, no isolates of were found. The study improves our understanding of the species associated with several disease symptoms on citrus plants, and provides useful information for effective disease management.
Preliminary study on fungal diversity in US soybean based on high throughput sequencing and culture methodology
Endophytic Phomopsis species: host range and implications for diversity estimates
Foliar endophyte assemblages of teak trees growing in dry deciduous and moist deciduous forests of Nilgiri Biosphere Reserve were compared. A species of Phomopsis dominated the endophyte assemblages of teak, irrespective of the location of the host trees. Internal transcribed spacer sequence analysis of 11 different Phomopsis isolates (ten from teak and one from Cassia fistula) showed that they fall into two groups, which are separated by a relatively long branch that is strongly supported. The results showed that this fungus is not host restricted and that it continues to survive as a saprotroph in teak leaf, possibly by exploiting senescent leaves as well as the litter. Although the endophyte assemblage of a teak tree growing about 500 km from the forests was also dominated by a Phomopsis sp., it separated into a different group based on internal transcribed spacer sequence analysis. Our results with an endophytic Phomopsis sp. reinforce the earlier conclusions reached by others for pathogenic Phomopsis sp., i.e., that this fungus is not host specific, and the species concept of Phomopsis needs to be redefined.
Diaporthe pseudolongicolla: The new pathogen on soybean seed in Serbia
Polymorphisms in nuclear rDNA and mtDNA reveal the polyphyletic nature of isolates of Phomopsis pathogenic to sunflower and a tight monophyletic clade of defined geographic origin
DOI:10.1017/S0953756204009372 URL [本文引用: 2]
The neighbor-joining method: a new method for reconstructing phylogenetic trees
DOI:10.1093/oxfordjournals.molbev.a040454
PMID:3447015
[本文引用: 1]

A new method called the neighbor-joining method is proposed for reconstructing phylogenetic trees from evolutionary distance data. The principle of this method is to find pairs of operational taxonomic units (OTUs [= neighbors]) that minimize the total branch length at each stage of clustering of OTUs starting with a starlike tree. The branch lengths as well as the topology of a parsimonious tree can quickly be obtained by using this method. Using computer simulation, we studied the efficiency of this method in obtaining the correct unrooted tree in comparison with that of five other tree-making methods: the unweighted pair group method of analysis, Farris's method, Sattath and Tversky's method, Li's method, and Tateno et al.'s modified Farris method. The new, neighbor-joining method and Sattath and Tversky's method are shown to be generally better than the other methods.
Primers for mating-type diagnosis in Diaporthe and Phomopsis: their use in teleomorph induction in vitro and biological species definition
DOI:10.1016/j.funbio.2010.01.007
PMID:20943136
[本文引用: 2]

Sexual reproduction in ascomycete fungi is governed by the mating-type (MAT) locus. The MAT loci of Diaporthe and its Phomopsis anamorphs differ in only one gene: MAT1-1-1 in mating-type MAT1-1 and MAT1-2-1 in mating-type MAT1-2. In order to diagnose mating-types in Diaporthe and Phomopsis and evaluate their usefulness in teleomorph induction in vitro and biological species delimitation, we designed primers that amplify part of the MAT1-1-1 and MAT1-2-1 genes. MAT phylogenies were generated and compared with ITS and EF1-α phylograms. Species recognised in the EF1-α phylogeny corresponded directly with those determined in the MAT phylogenies. ITS was shown to be highly variable resulting in a large number of phylogenetic species that were discordant with MAT and EF1-α species. Mating experiments were conducted to evaluate the existence of reproductive barriers between some isolates, and their anamorphic morphologies were compared. The primers proved to be useful in the mating-type diagnosis of isolates, selection of compatible mating pairs, and in the assessment of biological species boundaries.Copyright © 2010 The British Mycological Society. Published by Elsevier Ltd. All rights reserved.
Resolving the Diaporthe species occurring on soybean in Croatia
DOI:10.3767/003158511X603719
PMID:22403474
[本文引用: 5]

Diaporthe (anamorph = Phomopsis) species are plant pathogens and endophytes on a wide range of hosts including economically important crops. At least four Diaporthe taxa occur on soybean and they are responsible for serious diseases and significant yield losses. Although several studies have extensively described the culture and morphological characters of these pathogens, their taxonomy has not been fully resolved. Diaporthe and Phomopsis isolates were obtained from soybean and other plant hosts throughout Croatia. Phylogenetic relationships were determined through analyses of partial translation elongation factor 1-alpha (EF1-α) gene and ITS nrDNA sequence data. By combining morphological and molecular data, four species could be distinguished on soybeans in Croatia. Diaporthe phaseolorum is described in this study and its synonyms are discussed. Diaporthe phaseolorum var. caulivora is raised to species status and the name Diaporthe caulivora is introduced to accommodate it. A species previously known as Phomopsis sp. 9 from earlier studies on sunflower, grapevine, rooibos and hydrangea is reported for the first time on soybean, and is formally described as Diaporthe novem. The well-known soybean pathogen Phomopsis longicolla was also collected in the present study and was transferred to Diaporthe longicolla comb. nov. The presence of these species on herbaceous hosts raises once more the relevance of weeds as reservoirs for pathogens of economically important plants.
MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods
DOI:10.1093/molbev/msr121 URL [本文引用: 1]
CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice
DOI:10.1093/nar/22.22.4673
PMID:7984417
[本文引用: 1]

The sensitivity of the commonly used progressive multiple sequence alignment method has been greatly improved for the alignment of divergent protein sequences. Firstly, individual weights are assigned to each sequence in a partial alignment in order to down-weight near-duplicate sequences and up-weight the most divergent ones. Secondly, amino acid substitution matrices are varied at different alignment stages according to the divergence of the sequences to be aligned. Thirdly, residue-specific gap penalties and locally reduced gap penalties in hydrophilic regions encourage new gaps in potential loop regions rather than regular secondary structure. Fourthly, positions in early alignments where gaps have been opened receive locally reduced gap penalties to encourage the opening up of new gaps at these positions. These modifications are incorporated into a new program, CLUSTAL W which is freely available.
Diaporthe novem isolated from sunflower (Helianthus annuus) and other crop and weed hosts in Australia
DOI:10.1007/s10658-018-1515-7 URL [本文引用: 2]
The genus Phomopsis: biology, applications, species concepts and names of common phytopathogens
DOI:10.1007/s13225-011-0126-9 URL [本文引用: 1]
Reassessment of Phomopsis species on grapevines
DOI:10.1071/AP04072 URL [本文引用: 3]
Characterization of Phomopsis spp. associated with die-back of rooibos (Aspalathus linearis) in South Africa
DOI:10.3114/sim.55.1.65 URL [本文引用: 4]
Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics