中国块菌Tuber sinense K. Tao & B. Liu隶属于子囊菌门Ascomycota、子囊菌纲Ascomycetes、盘菌亚纲Pezizomycetidae、盘菌目Pezizales、块菌科Tuberaceae、块菌属Tuber F.H. Wigg,是一种地下生的大型真菌,别名猪拱菌、无娘果等。中国块菌子囊果呈不规则的球形、半球形或块状,其表皮上有棕色或黑褐色的疣突,成熟的块菌切面呈暗褐色或黑色,具有白色的大理石纹样(张世奇和阚建全 2011;清源等 2015;游金坤等 2015;卯晓岚 2000)。中国块菌主要生长于针、阔叶混交林下,与松科、壳斗科等植物共生,为外共生菌根真菌,因其共生机理研究薄弱,目前尚不能人工培养(陈应龙 2001;韩省华 2010)。块菌香气特殊,因此其化学成分的研究主要集中于挥发性风味物质的定性及定量分析,在挥发性风味成分中含量较多的是1-辛烯-3-醇、3-辛醇、2-辛烯-1-醇、1-辛烯-3-酮等八碳化合物(Culleré et al. 2012;李小林等 2015;Splivallo & Ebeler 2015;李杨等 2021)。近年的研究表明,块菌提取物具有抗炎、抗肿瘤、抗氧化、抗病毒、抑菌、保肝护肝等多种生理活性(Janakat et al. 2005;Al-Laith 2010;Patel 2012;Friedman 2015;呼鑫荣等 2017;范勇 2020),因此被称为预防疾病的“黑色钻石”。
中国块菌为云南的名贵野生菌,其广泛分布于滇东、滇西、滇东南地区,如禄劝、武定、禄丰、楚雄、大理、迪庆、文山等地,且大多发现于云南松Pinus yunnanensis Franch林下(薛燕等 2020),与云南松共生。
代谢组学技术是近年依赖核磁共振(1H-NMR)、气相质谱(GC-MS)、液质质谱(LC-MS)3大平台及日趋完善的代谢数据库快速发展起来的一种高通量分析技术,其以代谢组群指标分析为基础,可对生物的代谢成分进行无差别、整体、全面的分析。目前已广泛用于动物、植物的研究(王小娟等 2010;李静等2017;齐彦爽等2019),在菌物研究中报道尚少。因此本研究以采自云南松林下的新鲜块菌成熟子实体为研究材料,运用1H-NMR及GC-MS两种技术,对中国块菌及其共生云南松的小分子代谢产物进行了初步的研究,并对其差异物质及代谢通路进行了分析,旨在为探明中国块菌与其共生松树的共生机理和今后中国块菌相关药物的开发及人工培养的研究提供了理论依据。
1 材料与方法
1.1 材料
1.1.1 供试材料
新鲜的中国块菌子实体(图1)及松根:取自云南省禄劝县新华村;块菌经昆明食用菌研究所鉴定为中国块菌Tuber sinense,标本号为YNTuber-1;松根为云南松Pinus yunnanensis的细根,标本号为YNpinus-6;标本皆保存于云南大学生命科学学院植物标本馆。
图1
1.1.2 主要试剂和仪器
D2O (99.9%,TSP,0.05%);甲醇;甲氧胺盐酸盐(20 mg/mL:准确称取200 mg的甲氧胺盐酸盐,加入10 mL吡啶中,充分溶解,现配现用);吡啶;99% BSTFA+1% TMCS (99%双三甲基硅烷基三氟乙酰胺+1%三甲基氯硅烷);核糖醇。600兆核磁共振仪(Bruker 600-MHz AVANCE Ⅲ NMR Spectrometer,德国布鲁克公司);PTFE针头过滤器(0.22 μm,天津市津腾实验设备有限公司);气相色谱-质谱联用仪(HP6890GC/5973MS,美国Agilent Technologies公司)等。
1.2 样品制备
1.2.1 中国块菌
在无菌条件下分离块菌无菌子实体组织块,切碎,加入液氮,迅速将组织块研磨成粉末状,然后放入5 mL无菌冻存管中并置于-80 ℃超低温冰箱冷藏6 h以上,再进行真空冷冻干燥48 h至水分完全去除,用胶带密封管口后放入加有干燥剂的塑料袋中,扎紧袋口,于-80 ℃冷藏备用。
1.2.2 松根
将新鲜的松树细根用无菌水洗净、晾干,使用粉碎机将其打碎;加入液氮,迅速研磨成粉末状。其余步骤同上。
1.3 1H-NMR分析
1.3.1 1H-NMR提取溶剂的选择
本实验室前期通过水、甲醇、丙酮、氯仿、水与甲醇(1:1)、水与丙酮(1:1)、水与氯仿(1:1)、水+氯仿+甲醇(1:2:1) 8种溶剂的初筛实验表明,水提取出来的化合物种类较多,所以本实验室1H-NMR用水作为提取溶剂。具体提取方法见1.3.2,加入的8种溶剂分别为1 mL D2O、1 mL MeOD、1 mL C3D6O、1mL CDCl3、500 μL MeOD+ 500 μL D2O、500 μL C3D6O+ 500 μL D2O、500 μL CDCl3+ 500 μL D2O、1 mL CDCl3+ 500 μL D2O+ 500 μL MeOD。
1.3.2 1H-NMR样品制备
1.3.3 1H-NMR数据采集
测定温度为25 ℃,1H核的共振频率为600.13 MHz,采用压制水峰的noesypprld序列,谱宽为8 000 Hz,采样点数为32 K,信号扫描累加64次,数据点65 536,均以0.05% TSP为内标。手动进行相位、基线以及峰校正。所有的FID设定线宽因子为0.3 Hz。
1.3.4 1H-NMR数据定性分析
将1H-NMR自由感应衰减(free induction decay,FID)信号导入到Chenomx NMR suit (version 8.6,Chenomx,Edmonton,Canda)软件中,自动进行傅里叶转换,调整相位,校正基线。以TSP 峰(δ 0.0)作为全部谱图化学位移的基准,并对其进行反转卷积操作,调整谱图峰型(CSI)。根据1H-NMR谱图中信号的相关信息(如化学位移、峰型、半峰宽、耦合裂分等),结合Chenomx自带数据库对样本谱图的信号逐一比对分析,再结合核磁共振氢谱化学位移的3个区域:芳香区(δ 10.00-5.50)、糖区(δ 5.50-3.50)和氨基酸区(δ 3.50-0.00)对代谢产物进行指认。
1.3.5 1H-NMR相对定量分析
采用MestReNova (version 9.0,Mestrelab Research,Santiago de Compostella,Spain)对核磁图谱进行傅里叶转换并手动调整相位、基线,同时以TSP的化学位移(δ=0.00)进行化学位移校正。以δ 0.04积分段对化学位移区间δ 0.5-9.9进行分段积分,水相核磁图谱中去除水峰δ 4.70-5.02积分区间。分段积分值进行峰面积归一化处理,使数据集中在0-1内,保存在Excel文档,用于后续多元统计分析。
1.4 GC-MS分析
1.4.1 GC-MS样品制备
分别称取80 mg的块菌及松根冻干粉于2 mL离心管中(每个样重复称取4管),迅速加入1.5 mL预冷的混合溶剂(甲醇:三氯甲烷:水=1:1:1),将其旋涡震荡2 min后,于冰浴中进行45 KHz超声波辅助萃取1 h;取出,于4 ℃、13 000 r/min下离心10 min;将甲醇/水相提取物(400 µL)置于新的2 mL离心管中,加入100 µL 20 mg/mL的核糖醇作为内标,混匀后用真空浓缩仪将其完全蒸干;接着进行衍生化处理:先加入100 μL盐酸羟胺吡啶溶液(将盐酸羟胺溶于吡啶中,浓度为20 mg/mL),旋涡振荡2 min后,置于烘箱中37 ℃温浴120 min,再加入100 μL衍生化试剂(99% BSTFA+1% TMCS),密封后于37 ℃反应90 min,用1 mL注射器吸取液体,经0.22 μm聚四氟乙烯(PTFE)超滤膜过滤至气相小瓶中,待上机检测(孙时伟 2015)。
1.4.2 GC-MS上样条件
色谱分离条件:HP-5 MS石英毛细管柱(30 m×0.25 mm×0.25 mm);柱温起始温度150 ℃,以2 ℃/min程序升温至200 ℃,再5 ℃/min升温至280 ℃,保持20 min;柱流量为1.0 mL/min;进样口温度为250 ℃;柱前压100 kPa;进样量1.0 mL;分流比2:1;载气为高纯氦气。质谱条件:电离方式为EI;电子能量为70 eV;传输线温度为250 ℃;离子源温度为230 ℃;四极杆温度为150 ℃;质量范围为m/z 35-500。
1.4.3 谱图预处理
将所有的GC-MS原始数据(Raw格式)通过Agilent工作站导出为net CDF格式。将net CDF格式的GC-MS图谱导入XCMS-online进行峰提取、峰对齐、切割、滤噪代谢特征提取等预处理,去除柱流失、样品制备过程中的杂质峰等外界因素的干扰后,导入Excel中对数据进行归一化。
1.4.4 代谢产物的定性和定量
原始谱图背景扣除,保留时间校正和AMDIS软件解卷积分(除噪、峰匹配)操作再结合NIST (2014),PBM标准谱库,HMDB数据库(http://www.hmdb.ca/),标准品对代谢物逐一进行检索和定性。用匹配物质的峰面积值/内标峰面积值来表征该物质的含量,做相对定量分析,并用TBtools软件进行含量热图绘制。
1.5 差异代谢物分析
将1H-NMR和GC-MS积分数据分别矩阵导入SIMCA-P 14.1 (Umetrics,Umea,Sweden)软件,对数据进行中心化和规格化之后进行主成分分析(principal component analysis,PCA)和最小二乘法显著性分析(PLS-DA)来观察样本之间的总体分布和区分各组样本代谢轮廓的总体差异,通过正交偏最小二乘法判别分析(orthogonal PLS-DA,OPLS-DA)中变量权重值(variable importance in projection,VIP)大于1的组分和相关系数标注的载荷图中|P|>0.02和|P(corr)|>0.5的区域来确定差异变量。对差异变量进行定性和内标归一化定量后,采用统计分析软件SPSS 25进行方差分析,显著性分析采用t检验,其中P值小于0.05的组分被认为是具有显著性差异物质,P值小于0.01的组分认为是具有极显著性差异的物质。
1.6 代谢通路分析
将所筛选出的差异代谢物在Metaboanalyst 4.0平台(
2 结果与分析
2.1 核磁共振(1H-NMR)
表1 中国块菌子实体与云南松松根的1H-NMR峰指认
Table 1
| 序号 No. | 化合物名称 Compound | 化学位移 δH | 相对峰面积 Relative peak area | |
|---|---|---|---|---|
| SL | SG | |||
| 1 | Alloisoleucine 别异亮氨酸 | 0.960 (d, J=6 Hz); 0.931 (t, J=7.2 Hz) | 0.461±0.068 | 0.069±0.06 |
| 2 | Valine 缬氨酸 | 1.015 (d,J=7.2 Hz); 0.994 (d, J=7.2 Hz) | 0.355±0.064 | |
| 3 | Isobutyrate 异丁酸 | 1.047 (d, J=6.6 Hz) | 0.245±0.036 | |
| 4 | Ethanol 乙醇 | 1.186 (t, J=7.2 Hz) | 0.045±0.005 | |
| 5 | Cholate 胆酸盐 | 1.288 (m) | 0.886±0.203 | |
| 6 | Threonine 苏氨酸 | 1.333 (d, J=6.6 Hz); 3.579 (d, J=7.2 Hz) | 3.346±0.256 | 0.275±0.024 |
| 7 | Alanine 丙氨酸 | 1.480 (d, J=7.2 Hz) | 1.125±0.478 | 0.002±0.001 |
| 8 | Citrulline 瓜氨酸 | 1.538 (m); 1.556 (m) | 0.359±0.031 | |
| 9 | Arginine 精氨酸 | 1.934 (m); 1.905 (m); 1.731 (m); 1.649 (m) | 2.918±0.728 | |
| 10 | Tiglylglycine 虎甘氨酸 | 1.838 (s); 1.783 (d, J=3 Hz) | 2.612±0.773 | |
| 11 | 5-Hydroxylysine 5-羟基赖氨酸 | 3.166 (m); 3.158 (m) | 1.252±0.224 | |
| 12 | Glutamine 谷氨酰胺 | 2.464 (m); 2.437 (m); 2.156 (m); 2.117 (m) | 3.266±3.266 | |
| 13 | 4-Aminobutyrate 4-氨基丁酸 | 2.302 (t, J=7.8 Hz) | 0.219±0.023 | |
| 14 | Glutamate 谷氨酸 | 2.373 (m); 2.340 (m) | 1.033±0.366 | |
| 15 | Isocitrate 异柠檬酸盐 | 2.548 (m); 2.494 (m) | 1.751±0.505 | |
| 16 | Malate 苹果酸 | 2.681 (dd, J1=3, J2=15 Hz) | 0.720±0.221 | |
| 17 | Cystathionine 胱硫醚 | 2.738 (m) | 0.684±0.241 | |
| 18 | Asparagine 天冬酰胺 | 2.959 (dd, J1=4.2, J2=16.8 Hz) | 0.197±0.045 | |
| 19 | Lysine 赖氨酸 | 3.015 (t, J=7.8 Hz) | 0.224±0.016 | |
| 20 | Saccharopine 酵母氨酸 | 3.076 (m); 3.044 (m) | 0.297±0.044 | |
| 21 | Homocitrulline 高瓜氨酸 | 3.107 (m) | 0.228±0.058 | |
| 22 | Acetylcholine 乙酰胆碱 | 3.207 (s) | 0.566±0.175 | |
| 23 | Betaine 甜菜碱 | 3.235 (s) | 2.312±0.278 | 0.073±0.008 |
| 24 | Glucose-6-phosphate 6-磷酸葡萄糖 | 3.269 (t, J=6 Hz) | 1.370±0.407 | |
| 25 | Cellobiose 纤维二糖 | 3.742 (m); 3.725 (m); 3.335 (t, J=9.6 Hz) | 3.562±0.357 | |
| 26 | Glucose 葡萄糖 | 5.236 (d, J=3.6 Hz); 4.650 (d, J=7.8 Hz); 3.829 (m); 3.536 (d, J=3Hz); 3.492 (m); 3.469 (m); 3.413 (m); 3.403 (m) | 6.611±0.781 | 0.240±0.053 |
| 27 | Glycine 甘氨酸 | 3.554 (s) | 0.795±0.063 | 0.766±0.064 |
| 28 | Theophylline 茶碱 | 3.564 (s) | 0.769±0.144 | |
| 29 | Sarcosine 肌氨酸 | 3.607 (s) | 1.588±0.177 | 0.046±0.004 |
| 30 | Mannose 甘露糖 | 3.655 (m); 3.652 (m) | 9.024±0.430 | |
| 31 | Ethyleneglycol 乙二醇 | 3.663 (s) | 0.652±0.136 | |
| 32 | 2-Aminobutyrate 2-氨基丁酸 | 3.682 (t, J=6.6 Hz) | 4.087±0.193 | |
| 33 | Sucrose 蔗糖 | 3.825 (m); 3.781 (m); 3.774 (m) | 8.715±0.556 | |
| 34 | Mannitol 甘露醇 | 3.873 (dd, J1=2.4, J2=12 Hz) | 3.354±0.679 | 1.623±0.149 |
| 35 | 2-Phosphoglycerate 2-磷酸甘油酸 | 4.503 (m) | 0.316±0.064 | |
| 36 | Epicatechin 表儿茶素 | 5.026 (s) | 0.007±0.007 | |
| 37 | Fumarate 延胡索酸 | 6.524 (s) | 0.025±0.008 | 0.117±0.006 |
| 38 | Tyrosine 酪氨酸 | 6.897 (d, J=8.4 Hz) | 0.038±0.006 | |
| 39 | Phenylalanine 苯丙氨酸 | 7.430 (t, J=7.2 Hz); 7.378 (t, J=7.2 Hz); 7.331 (d, J=7.8 Hz) | 0.120±0.065 | 1.555±0.117 |
| 40 | Pantothenate 泛酸盐 | 8.006 (s) | 0.007±0.002 | |
| 41 | Proline 脯氨酸 | 1.97 (m); 2.02 (m); 2.05 (m); 2.36 (m); 3.34 (m) | 0.357±0.066 | |
| 42 | Glycylproline 甘氨酰脯氨酸 | 3.569 (m); 3.537 (m) | 1.517±0.173 | |
| 43 | Asparate 天冬氨酸 | 2.82 (dd, J1=6, J2=12 Hz) | 0.948±0.082 | |
| 44 | Fructose 果糖 | 4.099 (s); 3.808 (d, J=10.2 Hz); 3.793 (d, J=9 Hz) | 0.508±0.047 | |
| 45 | Trehalose 海藻糖 | 3.45 (t, J=9.0 Hz); 5.20 (d, J=3.6 Hz) | 0.302±0.044 | |
| 46 | Fucose 岩藻糖 | 1.241 (d, J=7.8 Hz); 1.207 (d, J=6 Hz) | 0.537±0.045 | |
| 47 | Arabinose 阿拉伯糖 | 4.516 (d, J=8.4 Hz) | 0.127±0.021 | |
| 48 | Arabinitol 阿拉伯糖醇 | 3.841 (dd, J1=3, J2=13.8 Hz); 3.667 (m); 3.641 (m) | 0.962±0.078 | |
| 49 | myo-Inositol 肌醇 | 3.623 (d, J=8.4 Hz) | 0.368±0.042 | |
| 50 | 4-Carboxyglutamate 4-羧基谷氨酸盐 | 3.707 (dd, J1=2.4, J2=11.4 Hz) | 1.706±0.146 | |
| 51 | Gluconate 葡萄糖酸盐 | 4.033 (s) | 0.394±0.026 | |
| 52 | Choline 胆碱 | 3.21 (s) | 0.011±0.008 | |
| 53 | O-Phosphocholine 磷酸胆碱 | 4.148 (s) | 0.438±0.031 | |
| 54 | 1,3-Dihydroxyacetone 1,3-二羟基丙酮 | 4.409 (s) | 0.257±0.022 | |
| 55 | Acetone 丙酮 | 2.225 (s) | 0.111±0.011 | |
注:SL:中国块菌;SG:松根
Note: SL: Tuber sinense; SG: Pinus yunnanensis root.
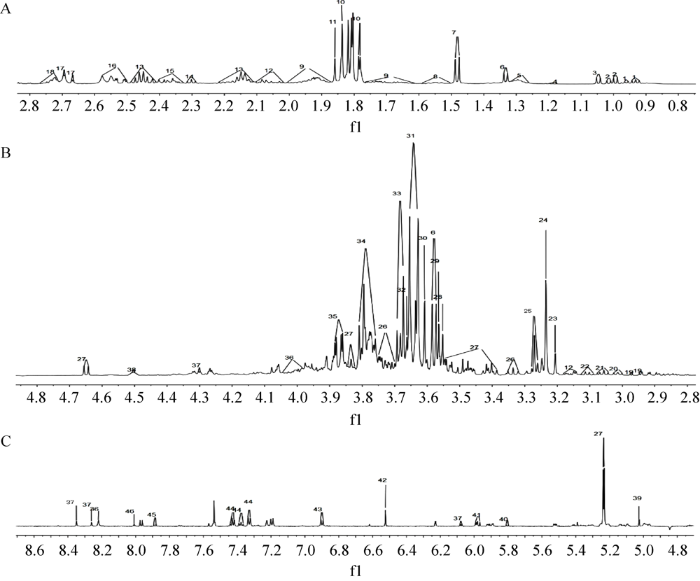
图2
图2
中国块菌子实体水溶性提取物1H-NMR指认图谱
Fig. 2
1H-NMR identified spectra of water-soluble extract of Tuber sinense fruiting bodies. A: δ 0.8-2.8; B: δ 2.8-4.8; C: δ 4.8-8.6.
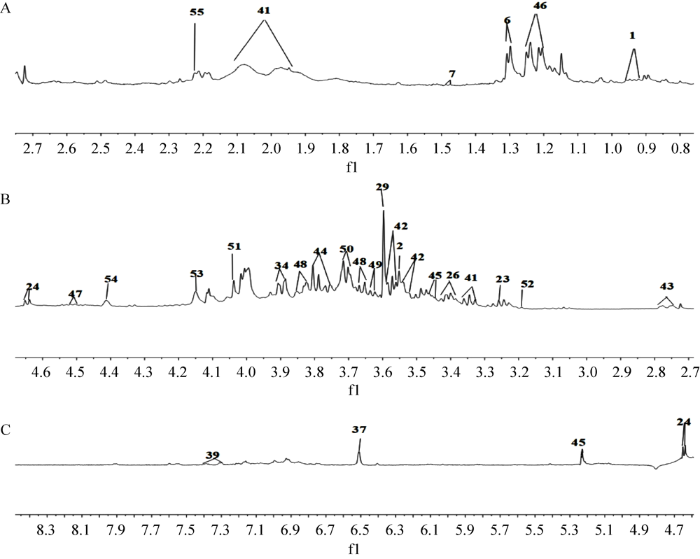
图3
图3
松根水溶性提取物1H-NMR指认图谱
Fig. 3
1H-NMR identified spectra of water-soluble extract of Pinus yunnanensis root. A: δ 0.8-2.7; B: δ 2.7-4.6; C: δ 4.6-8.5.
2.2 气相色谱质谱联用(GC-MS)
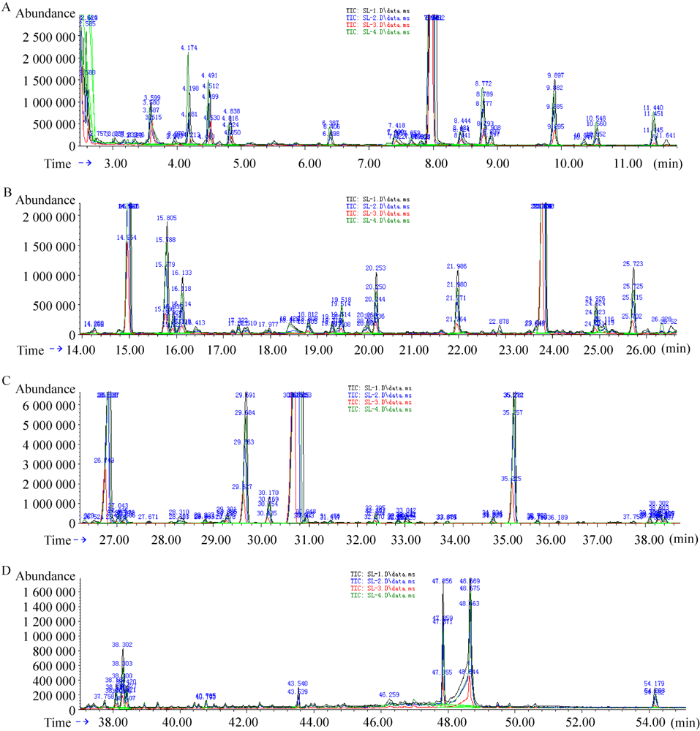
图4
图4
中国块菌子实体的GC-MS总离子流分析图谱
Fig. 4
TIC of GC-MS analysis map of Tuber sinense fruiting bodies. A: 0-13 min; B: 13-26 min; C: 26-38 min; D: 38-55 min.
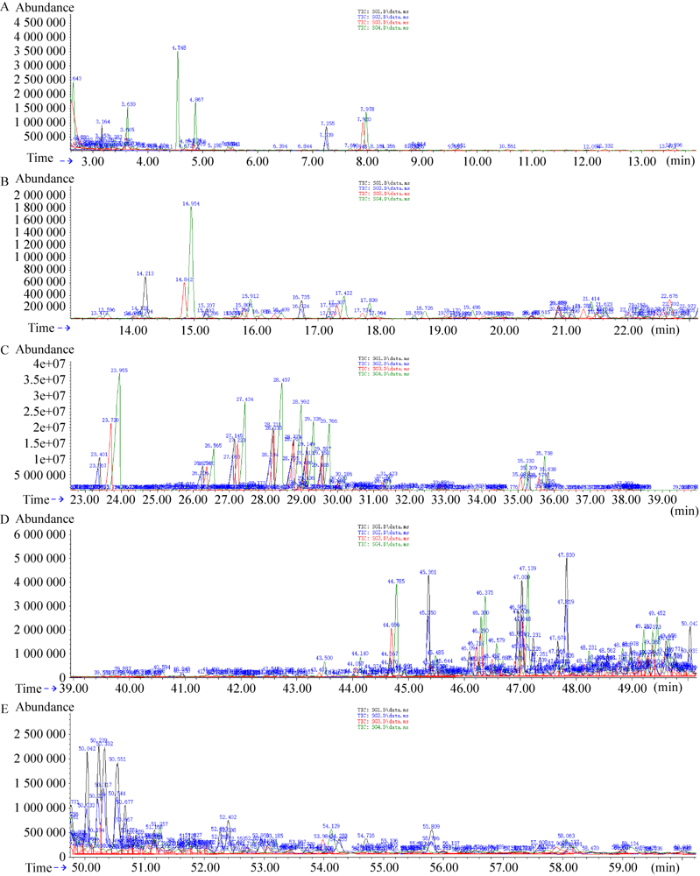
图5
图5
松根的GC-MS总离子流分析图谱
Fig. 5
TIC of GC-MS analysis map of Pinus yunnanensis root. A: 0-13 min; B: 13-23 min; C: 26-39 min; D: 39-50 min; E: 50-60 min.
表2 中国块菌子实体及松根的GC-MS峰的指认
Table 2
| 序号 No. | 保留时间 RT (min) | 代谢产物名称 Metabolites | 分子式 Formula | 分子量 m/z | 相对峰面积 Relative peak area | |
|---|---|---|---|---|---|---|
| SL | SG | |||||
| 1 | 3.037 | Propanoic acid丙酸 | C3H6O2 | 74.08 | 0.092±0.030 | 0.017±0.006 |
| 2 | 3.97 | L-Valine缬氨酸 | C5H11NO2 | 117.15 | 0.029±0.013 | |
| 3 | 4.198 | l-Alanine丙氨酸 | C3H7NO2 | 89.09 | 0.059±0.032 | |
| 4 | 4.44 | Glycine甘氨酸 | C2H5NO2 | 75.07 | 0.089±0.019 | |
| 5 | 4.838 | Ethanedioic acid乙二酸 | H₂C₂O₄ | 90.03 | 0.033±0.005 | 0.013±0.006 |
| 6 | 7.418 | Serine丝氨酸 | C3H7NO3 | 105.09 | 0.053±0.013 | |
| 7 | 7.821 | L-Leucine亮氨酸 | C6H13NO2 | 131.17 | 0.006±0.003 | |
| 8 | 8.44 | l-Threonine苏氨酸 | C4H9NO3 | 119.12 | 0.066±0.021 | |
| 9 | 8.908 | Butanedioic acid琥珀酸 | C4H6O4 | 118.09 | 0.490±0.041 | 0.024±0.008 |
| 10 | 9.897 | 2-Butenedioic acid柠康酸 | C5H6O4 | 130.1 | 0.092±0.010 | |
| 11 | 11.641 | l-Aspartic acid天门冬氨酸 | C4H7NO4 | 133.10 | 0.008±0.004 | |
| 12 | 15.805 | L-Proline脯氨酸 | C5H9NO2 | 115.13 | 0.152±0.013 | |
| 13 | 15.935 | Meso-Erythritol赤藓糖醇 | C4H10O4 | 122.12 | 0.046±0.019 | |
| 14 | 17.51 | 3-Pyridinecarboxylic acid烟酸 | C6H5NO2 | 123.11 | 0.014±0.006 | |
| 15 | 17.977 | Pentanedioic acid戊二酸 | C5H8O4 | 132.11 | 0.005±0.002 | |
| 16 | 18.422 | L-Asparagine天冬酰胺 | C4H8N2O3 | 132.12 | 0.035±0.018 | |
| 17 | 19.514 | Glutamic acid谷氨酸 | C5H9NO4 | 147.13 | 0.042±0.010 | |
| 18 | 25.723 | 2-Keto-l-gluconic acid | C6H10O7 | 194.14 | 0.079±0.005 | |
| 2-酮基-D-葡萄糖酸 | ||||||
| 19 | 26.857 | 1,2,3-Propanetricarboxylic acid | C6H8O6 | 176.12 | 1.741±0.864 | |
| 1,2,3-丙三羧酸 | ||||||
| 20 | 27.185 | Lysine赖氨酸 | C6H14N2O2 | 146.19 | 0.036±0.010 | |
| 21 | 28.837 | D-(-)-Fructose果糖 | C6H12O6 | 180.16 | 0.026±0.362 | 1.104±0.365 |
| 22 | 29.691 | d-Glucose葡萄糖 | C6H12O6 | 180.16 | 0.613±0.117 | 0.426±0.141 |
| 23 | 30.17 | D-Allose阿洛糖 | C6H12O6 | 180.15 | 0.111±2.784 | |
| 24 | 32.395 | Glucopyranose吡喃葡萄糖 | C6H12O6 | 180.16 | 0.043±0.025 | |
| 25 | 32.86 | D-Gluconic acid葡萄糖酸 | C6H12O7 | 196.16 | 0.012±0.006 | 0.015±0.005 |
| 26 | 33.042 | Hexadecanoic acid棕榈酸 | C16H32O2 | 256.42 | 0.032±0.005 | 0.032±0.020 |
| 27 | 38.103 | Octadecanoic acid硬脂酸 | C18H36O2 | 284.48 | 0.037±0.010 | 0.018±0.015 |
| 28 | 38.42 | Glucitol山梨醇 | C6H14O6 | 182.17 | 4.838±0.027 | 0.025±0.009 |
| 29 | 47.856 | D-(+)-Trehalose海藻糖 | C12H22O11 | 342.30 | 0.187±0.019 | 0.023±0.008 |
| 30 | 48.669 | D-Myo-Inisitol肌醇 | C6H12O6 | 180.16 | 1.262±0.004 | |
| 31 | 5.501 | Pentasiloxane十二甲基五硅氧烷 | C12H36O4Si5 | 384.84 | 0.001±0.001 | |
| 32 | 17.72 | L-Threonic acid苏糖酸 | C4H8O5 | 136.10 | 0.003±0.001 | |
| 33 | 21.288 | DL-Arabinose阿拉伯糖 | C5H10O5 | 150.13 | 0.009±0.003 | |
| 34 | 22.459 | Levoglucosan内醚糖 | C6H10O5 | 162.14 | 0.001±0.001 | |
| 35 | 22.57 | Xylitol木糖醇 | C5H12O5 | 152.15 | 0.004±0.001 | |
| 36 | 26.371 | 1-Cyclohexene-1-carboxylic acid | C7H10O2 | 126.15 | 0.302±0.101 | |
| 1-环己烯-1-甲酸 | ||||||
| 37 | 27.226 | D-Pinitol松醇 | C7H14O6 | 194.18 | 0.714±0.236 | |
| 38 | 30.003 | d-Mannose甘露糖 | C6H12O6 | 180.16 | 0.073±0.025 | |
| 39 | 30.447 | D-Mannitol甘露醇 | C6H14O6 | 182.17 | 0.022±0.007 | |
| 40 | 35.078 | Myo-Inositol肌醇 | C6H12O6 | 180.16 | 0.133±0.045 | |
| 41 | 35.709 | 2-Propenoic acid | C15H28O2 | 240.38 | 0.009±0.003 | |
| 正十一烷基甲基丙烯酸酯 | ||||||
| 42 | 44.695 | 3-α-Mannobiose甘露二糖 | C12H22O11 | 342.40 | 0.050±0.017 | |
| 43 | 46.29 | β-Gentiobiose龙胆二糖 | C12H22O11 | 342.30 | 0.052±0.017 | |
| 44 | 50.194 | Aucubin桃叶珊瑚苷 | C15H22O9 | 346.33 | 0.016±0.004 | |
| 45 | 44.057 | D-(+)-Cellobiose纤维二糖 | C12H22O11 | 342.30 | 0.006±0.004 | |
| 46 | 46.214 | α-D-Glucopyranoside吡喃葡萄糖苷 | C26H44O8 | 484.63 | 0.028±0.006 | |
注:SL:中国块菌;SG:松根
Note: SL: Tuber sinense; SG: Pinus yunnanensis root.
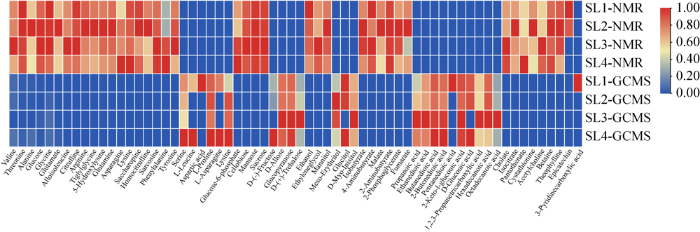
2.3 两种检测方法的比较
图6
图6
两种方法检测的中国块菌子实体的代谢产物及其含量
Fig. 6
Metabolites and their content of Tuber sinense fruit bodies detected by two methods.
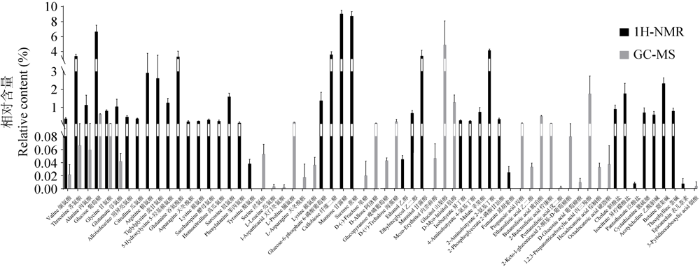
图7
图7
两种方法检测的中国块菌子实体的代谢产物及其含量比较
Fig. 7
Comparison of metabolites and their content of Tuber sinense fruit bodies detected by two methods.
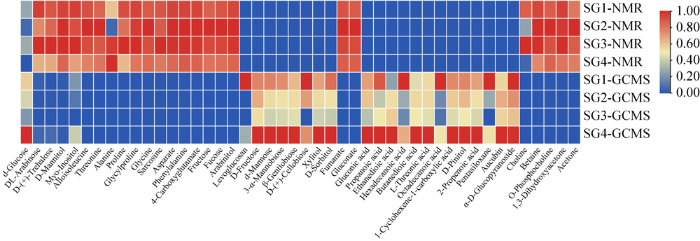
图8
图8
两种方法检测的云南松松根的代谢产物及其含量
Fig. 8
Metabolites and their content of Pinus yunnanensis roots detected by two methods.
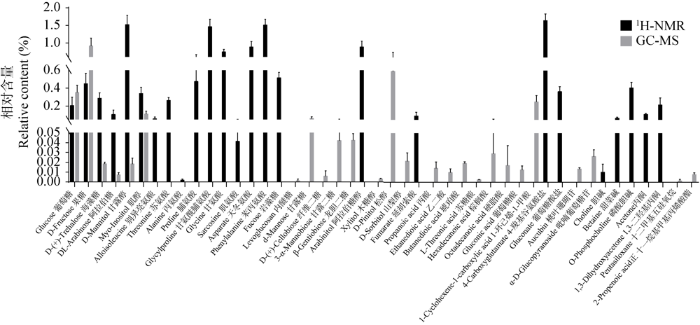
图9
图9
两种方法检测的云南松松根的代谢产物及其含量比较
Fig. 9
Comparison of metabolites and their content of Pinus yunnanensis roots detected by two methods.
2.4 中国块菌和松根的差异代谢物分析
2.4.1 主成分分析(PCA)
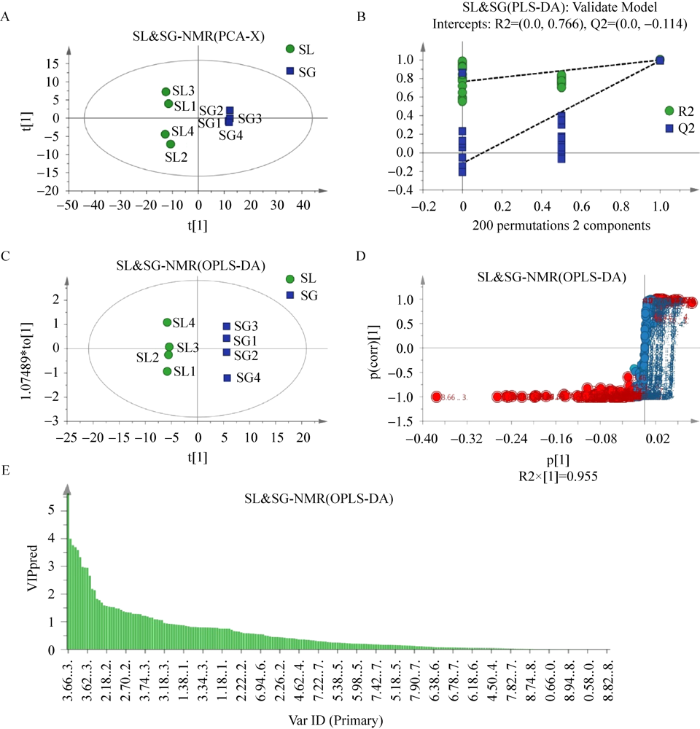
在主成分分析散点图10A中,块菌组内4个重复样SL1、SL3与SL2、SL4相对分散,但全部位于横轴的负半轴,松根组全部位于横轴的正半轴,分离趋势明显,且无异常值存在,说明两组数据间存在明显差异;PCA虽能反映不同样本之间的整体差别及数据原始的状态,但属于无监督的分析方法,在分析差异成分时不能忽略组内误差和消除随机误差,因此,需要采用有监督的正交偏最小二乘判别分析(OPLS-DA)来进一步确定比较组分之间的差异成分,OPLS-DA 分析必须以模型验证为基础(图10B),经过200次交叉验证,左端任何一次随机排列产生的R2、Q2均小于右端的原始值,Q2与纵轴交点为-0.114<0,验证结果显示模型有效,且说明两组之间的化学差异具有统计学意义;图10C为OPLS-DA散点图,OPLS模型验证参数P=5.75E-06<0.05,表征模型验证成立,两组样品得到最大分离,有效降低了组内差异;图10D为S-plots 图,图中组成“S”曲线的每一个点代表一个变量,其中表示一个变量的点距离原点越远,表明该变量对这组差异的置信水平越高,结合图E中VIP>1以及独立样本t检验P<0.05来筛选出具有显著性差异的代谢物。
图10
图10
基于1H-NMR技术的中国块菌和松根代谢产物的多元统计学分析 A:PCA得分图;B:置换模型验证;C:OPLS-DA得分图;D:S-Plots;E:VIP值
Fig. 10
Multivariate statistical analysis of metabolites of Tuber sinense fruit bodies and Pinus yunnanensis roots based on 1H-NMR technique. A: PCA score scatter plot; B: PLS-DA model validation diagram; C: OPLS scatter plot; D: S-Plot; E: VIP value.
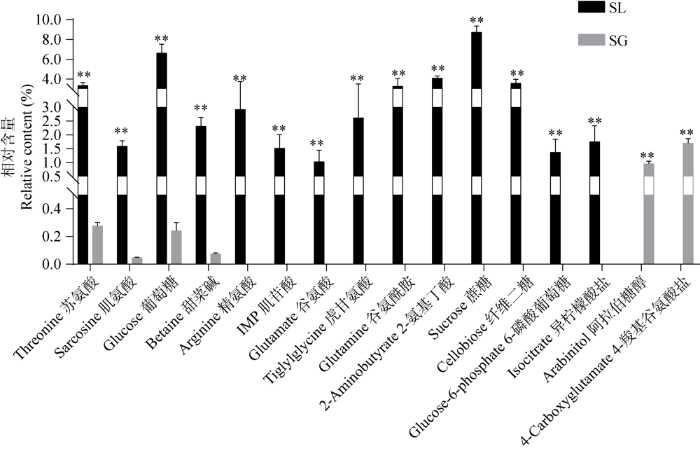
筛选出的差异代谢产物及含量见图11,有16个差异代谢产物。其中阿拉伯糖醇和4-羧基谷氨酸盐仅存在于松根中,精氨酸、肌苷、谷氨酸、虎甘氨酸、谷氨酰胺、2-氨基丁酸、蔗糖、纤维二糖、6-磷酸葡萄糖以及异柠檬酸盐为块菌特有,苏氨酸、肌氨酸、葡萄糖、甜菜碱4个物质,在块菌中极显著高于松根。
图11
图11
基于1H-NMR技术的中国块菌和松根之间差异代谢产物的相对含量 SL:中国块菌;SG:松根. ** P<0.01
Fig. 11
Relative content of differential metabolites of Tuber sinense fruit bodies and Pinus yunnanensis roots based on 1H-NMR technique. SL: Tuber sinense; SG: Pinus yunnanensis root. ** P<0.01.
2.4.2 基于GC-MS技术的块菌和松根的差异代谢物分析
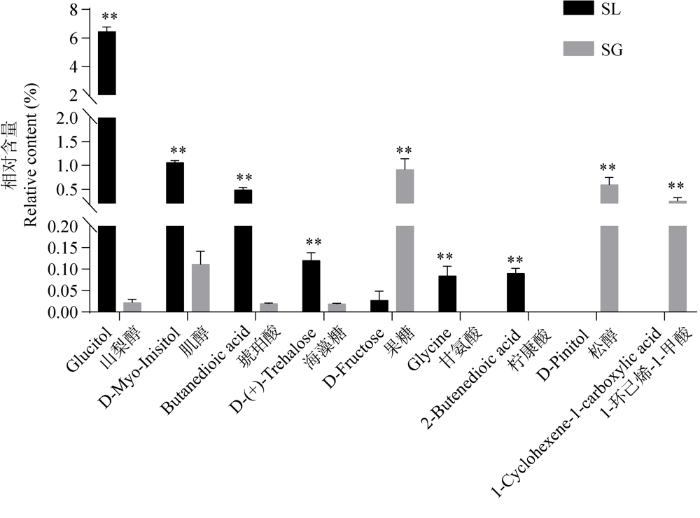
通过多元统计学分析表明,两组数据在第一主成分上有明显的分离,模型建立参数P=2.60E- 05<0.05,显示出模型具有较好的预测能力,可用于进一步的差异代谢产物分析。模型验证后筛选出的差异代谢产物及含量见图12,有9个差异代谢产物。松醇和1-环己烯-1-甲酸仅存在于松根中,甘氨酸和柠康酸仅存在于块菌中,除果糖以外,山梨醇、肌醇、琥珀酸、海藻糖在块菌中的含量显著高于松根。
图12
图12
基于GC-MS技术的中国块菌和松根之间差异代谢产物的相对含量 SL:中国块菌;SG:松根
Fig. 12
Relative content of differential metabolites of Tuber sinense fruit bodies and Pinus yunnanensis roots based on GCMS technique. SL: Tuber sinense; SG: Pinus yunnanensis root.
2.5 中国块菌和云南松松根之间差异代谢物的代谢通路分析
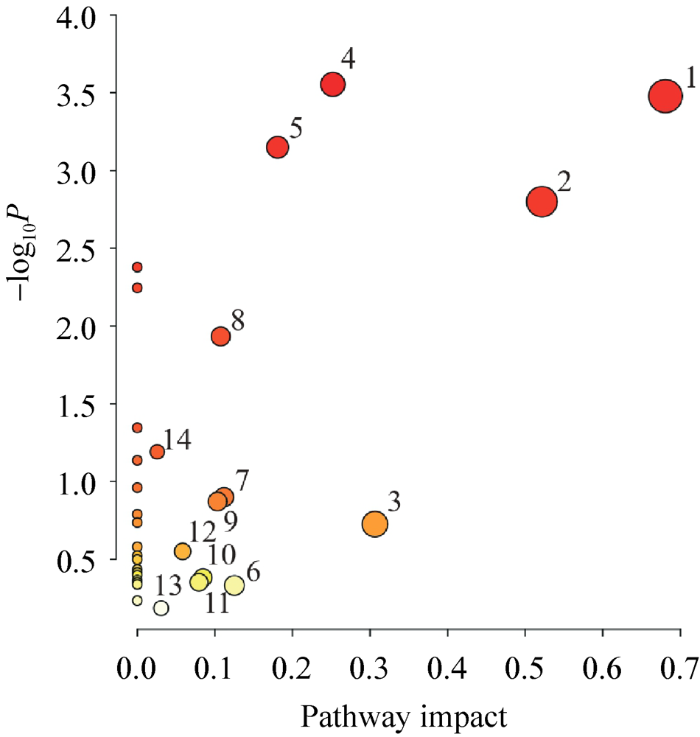
通过两种技术检测到的所有块菌与松根之间的差异代谢产物的代谢通路分析结果见图13。图中Y轴基于P值(来自通路富集分析),X轴基于代谢通路影响值(来自通路拓扑分析);节点颜色受P值影响,由浅到深,P值由大到小,节点半径为代谢通路影响值的体现,半径越大影响值越大。
图13
图13
中国块菌和松根之间差异代谢物的代谢通路分析 1:淀粉和蔗糖代谢;2:丙氨酸、天冬氨酸和谷氨酸代谢;3:甘氨酸、丝氨酸和苏氨酸的代谢;4:乙醛酸和二羧酸代谢;5:精氨酸生物合成;6:嘌呤代谢;7:精氨酸和脯氨酸的代谢;8:三羧酸循环(TCA循环);9:谷胱甘肽代谢;10:苯丙氨酸、酪氨酸和色氨酸的生物合成;11:甲烷代谢;12:硫代谢;13:半胱氨酸和蛋氨酸代谢;14:半乳糖代谢
Fig. 13
Metabolic pathway of differential metabolites between Tuber sinense fruit bodies and Pinus yunnanensis roots. 1: Starch and sucrose metabolism; 2: Alanine, aspartate and glutamate metabolism; 3: Glycine, serine and threonine metabolism; 4: Glyoxylate and dicarboxylate metabolism; 5: Arginine biosynthesis; 6: Purine metabolism; 7: Arginine and proline metabolism; 8: Citrate cycle (TCA cycle); 9: Glutathione metabolism; 10: Phenylalanine, tyrosine and tryptophan biosynthesis; 11: Methane metabolism; 12: Sulfur metabolism; 13: Cysteine and methionine metabolism; 14: Galactose metabolism.
本研究选择代谢通路影响值大于0作为筛选条件,综合比较得出块菌子实体与松根之间差异代谢产物涉及的相关代谢通路共14个,其中影响最大的代谢通路有5个,具体如下:淀粉和蔗糖代谢,丙氨酸、天冬氨酸和谷氨酸代谢,甘氨酸、丝氨酸和苏氨酸的代谢,乙醛酸和二羧酸代谢,精氨酸生物合成代谢,以淀粉和蔗糖代谢通路影响最大。
3 讨论
3.1 1H-NMR及GC-MS方法
核磁共振氢谱法(1H-NMR)具有无偏向性、样品处理简单且无损伤性、定量准确、重复性高和成本低等特性,但其缺点是,检测的动态范围较狭窄(Emwas 2015;朱航等 2006)。GC-MS具有较高的分辨率,可以对多个化合物同时进行分析和鉴定,且有成熟的标准参考谱库,通过比对标准谱库可以简单高效地对代谢产物进行定性分析,但其缺点是,对于难挥发性的代谢产物需要进行衍生化或甲酯化反应,增加了样品前处理的复杂程度(Farag et al. 2009;王斯婷等 2010;Fiehn 2016)。两种方法都见于大型真菌的代谢产物研究。
本研究同时利用1H-NMR和GC-MS两种方法对中国块菌及其共生的松根进行了小分子活性代谢物的检测,在块菌中分别指认出了40和30个物质,可见1H-NMR检测到的物质相对多于GC-MS。总体上两种方法互为补充,扩大了检测效果。但由于提取方法的多样性及数据库的完整性等,提取到的化合物不够全面,大部分峰谱尚难以识别,后续可以进一步结合LC-MS技术,进行更加系统性及完整性的分析。
3.2 中国块菌的代谢产物
在检测到的块菌代谢产物中,除了氨基酸、糖类和酸类外,还检测到几种活性物质如烟酸(维生素B3)、甜菜碱、茶碱及表儿茶素。烟酸是细胞代谢辅酶烟酰胺腺嘌呤二核苷酸(NAD+)和烟酰胺腺嘌呤二核酸磷(NADP+)的前体,其通过参与人体的脂质代谢、氧化过程和无氧分解过程,调控炎症反应、调节肠道稳态及维持肠道健康状态,还可治疗维持性血液透析患者高磷血症(高格 2019)。甜菜碱参与生物体内合成肉碱、肌酸等必需物质的系列化反应,是生物体内稳定高效的甲基供体,不仅可改善高脂饮食诱导的小鼠体重增加和胰岛素抵抗,并增强脂质和能量代谢,还可促进骨骼肌发育、提高人或小鼠的运动能力、缓解酒精性脂肪肝等(Du et al. 2018;何劲等 2020)。茶碱可非特异性地抑制环核苷酸磷酸二酯酶活性,进而有效增加细胞内环磷酸腺苷浓度水平,提高细胞内环磷酸腺苷/环磷酸鸟苷比值,抑制钙离子内流及促进钙离子外流,起到扩张支气管平滑肌的作用,在临床上主要用于治疗慢性阻塞性肺疾病和哮喘;茶碱还具有抗炎、调节免疫、抑制血小板活性的作用(李媛和林青 2013)。表儿茶素是一种分布广泛的天然植物黄烷醇化合物,研究表明表儿茶素具有抗氧化、调节免疫、预防心血管疾病、降脂降糖、降低胰岛素、抑菌、抗炎、保护神经等作用(童观珍等 2018)。这些有效活性成分为块菌保健药物的开发提供了理论依据。
3.3 中国块菌与松根的差异物质分析
中国块菌(云南黑松露)主要与松根共生,属于外生菌根真菌,菌根真菌与宿主植物通过菌根共生体互惠互益共同生长。菌根共生体是物质和信息传递的场所。
研究发现,在共生菌根形成过程中,宿主植物可通过分泌类黄酮来实现信号传导;外生菌根真菌则通过合成并分泌生长素和脂壳寡糖、释放倍半萜实现信号传递,这些物质进入植物根系细胞后,激活植物生长素信号通路和通用共生信号通路等相关通路,抑制植物根系生长并影响侧根和哈蒂氏网的形成(冯邦和杨祝良 2019)。
碳素同化及氮营养对菌根的合成具有至关重要的作用。菌根共生过程会产生重度的碳饥渴,促使高达30%的光合产物转移到菌根合成中,以供给菌根菌使用(Stitt 1991)。菌根最初的合成需要大量的氮源,已经证明了内聚合氨基酸影响共生真菌的侵染(Baum 2002)。
参考文献
Antioxidant components and antioxidant/antiradical activities of desert truffle (Tirmania nivea) from various Middle Eastern origins
DOI:10.1016/j.jfca.2009.07.005 URL [本文引用: 1]
The effects of nitrogen fertilization and soil properties on mycorrhizal formation of Salix viminalis
DOI:10.1016/S0378-1127(01)00470-4 URL [本文引用: 1]
Ecological studies on truffles (Tuber spp.)
Evaluation of gamma and electron-beam irradiation on the aromatic profile of black truffle (Tuber melanosporum) and summer truffle (Tuber aestivum)
DOI:10.1016/j.ifset.2011.09.003 URL [本文引用: 1]
Betaine supplementation enhances lipid metabolism and improves insulin resistance in mice fed a high-fat diet
DOI:10.3390/nu10020131 URL [本文引用: 1]
The strengths and weaknesses of NMR spectroscopy and mass spectrometry with particular focus on metabolomics research
Action principle of tuber polysaccharide improving sports fatigue
Comparative metabolite profiling and fingerprinting of medicinal licorice roots using a multiplex approach of GC-MS, LC-MS and 1D NMR techniques
Ectomycorrhizal symbioses: diversity of mycobionts and molecular mechanisms that entail the development of ectomycorrhizae
Metabolomics by gas chromatography-mass spectrometry: combined targeted and untargeted profiling
Chemistry, nutrition, and health-promoting properties of Hericium erinaceus (lion’s mane) mushroom fruiting bodies and mycelia and their bioactive compounds
A systematic review: niacin and nicotinamide for the treatment of hyperphosphatemia in maintenance hemodialysis patients
The black diamond “truffle” in edible fungi
Preliminary study on the effect of betaine on lipid deposition in muscle tissue of mice with leptin deficiency
Research progress on the composition and functional activity of truffles
Evolution of antibacterial activity of aqueous and methanolic extracts of the truffle Terfezia claveryi against Pseudomonas aeruginosa
To investigate the antibacterial activities of aqueous and methanolic extracts, as well as, partially purified proteins extracted from Terfezia claveryi aqueous, against Pseudomonas aeruginosa (P. aeruginosa).Five percent of the aqueous and methanolic extracts were added to growth medium of P. aeruginosa. The extract that caused growth inhibition (aqueous) was then partially purified using ammonium sulfate precipitation, gel chromatography and ion exchange chromatography. Antibacterial activities of the obtained fractions were assessed using agar-well diffusion test, and then all the results were compared with reference antibiotics. Excremental procedures were performed at the Department of Nutrition and Food Technology and the animal house of Jordan University of Science and Technology, Jordan during the year 2000.Five percent aqueous extract inhibited the growth of P. aeruginosa by 40.9%, while methanolic extract was ineffective. Partial purification of the aqueous extract using ammonium sulfate precipitation revealed that antimicrobial activity was within the second pellet (25-45%). This fraction was then subjected to gel permeation chromatography using Sephadex G-25. Peak one, of the 2 peaks obtained, possessed higher antimicrobial activity. Peak one was then subjected to ion exchange chromatography using DEAE Sephadex. Only peak one, of the 3 peaks obtained, showed a slight antimicrobial activity. Relative antimicrobial activities of these fractions were found to be superior to most of reference antibiotics used for comparison.Aqueous extract of the truffle Terfezia claveryi contains a potent antimicrobial agent that is protein in nature and may be used in the treatment of eye infections caused by P. aeruginosa.
Chemical comparison on different parts of Angelica sinensis radix based on NMR metabolomics
Analysis of volatile aroma components in different species of truffle in Huidong County by GC-MS
Research progress on chemical constituents and pharmacological effects of Tuber (Pezizales, Ascomycota)
Research progress and application of theophylline drugs
Chinese macrofungi
The 1H-NMR analysis of water soluble metabolites of three major growth and development stages of Xerocomus spadiceus
Food, health and agricultural importance of truffles: a review of current scientific literature
Chemical comparison of the stems and leaves of Tussilago farfara L. using NMR-based metabolomics
Application of electronic nose and tongue for discrimination of truffles with different processing technology
Sulfur volatiles of microbial origin are key contributors to human-sensed truffle aroma
DOI:10.1007/s00253-014-6360-9
PMID:25573471
[本文引用: 1]

Truffles are symbiotic fungi in high demand for the aroma of their fruiting bodies which are colonized by a diverse microbial flora. Specific sulfur containing volatiles (thiophene derivatives) characteristic of the white truffle Tuber borchii were recently shown to be derived from the bacterial community inhabiting truffle fruiting bodies. Our aim here was to investigate whether thiophene derivatives contributed to the human-sensed aroma of T. borchii. Furthermore, we questioned whether the concentration of thiophene volatiles was affected by freezing or whether it differed in truffles from distinct geographical origins. Gas chromatography-olfactometry (GC-O) analysis revealed that thiophene derivatives were major contributors to the aroma of T. borchii. Of four thiophene derivatives detected in this study, 3-methyl-4,5-dihydrothiophene was the most important one in terms of its contribution to the overall aroma. The relative concentration of thiophene derivatives was unaffected by freezing; however, it differed in samples collected in distinct geographical locations (Italy versus New Zealand). The causes of this variability might be differences in storage conditions and/or in bacterial community composition of the fruiting bodies; however, further work is needed to confirm these hypotheses. Overall, our results demonstrate that thiophene derivatives are major contributors to the human-sensed aroma of T. borchii.
Rising CO2 levels and their potential significance for carbon flow in photosynthetic cells
DOI:10.1111/pce.1991.14.issue-8 URL [本文引用: 1]
The preliminary construction and analysis on the metabolic profiling of three medicinal dendrobes
Advances in research on the distribution and pharmacological activities of epicatechin
Metabonomics and its analytical technique
Sample pretreatment method in metabolomics research based on NMR
Ecological climate suitability and geographical distribution of truffle in west Panxi area
Nutrient analysis of two Tuber huidongense from Yunnan
Aroma compound analysis of Tuber huidongense by GC-MS
NMR based metabonomics