Gerdemann & Trappe (1974)根据 AM 真菌孢子形成于产孢细胞末端这一特性,建立了巨孢囊霉属Gigaspora (内囊霉科Endogonaceae;接合菌门Zygomycota),包括5个种,极大巨孢囊霉Gigaspora gigantea为模式种,到1985年,该属共有18个物种(Becker & Hall 1976;Bhattacharjee & Mukerji 1980;Koske & Walker 1984, 1985)。Walker & Sanders (1986)根据孢子壁或发芽盾特征,建立了一个新属——盾巨孢囊霉属Scutellospora,含原巨孢囊霉属中的17个物种,美丽盾巨孢囊霉Scutellospora calospora为模式种。Morton & Benny (1990)再次根据孢子壁结构和发芽盾特征,建立了巨孢囊霉科Gigasporineae (球囊霉目Glomerales),含巨孢囊霉属和盾巨孢囊霉属。Schüßler & Walker (2004)利用核糖体小亚基(SSU)序列确立了多样孢囊霉目Diversisporales,并将巨孢囊霉科归入其中。目前,巨孢囊霉科包含8个属:裂盾囊霉属Racocetra、盾孢囊霉属Cetraspora、齿盾囊霉属Dentiscutata、巨孢囊霉属Gigaspora、盾巨孢囊霉属Scutellospora、内饰孢囊霉属Intraornatospora、类齿盾霉属Paradentiscutata和葱状囊霉属Bulbospora (王幼珊和刘润进 2017)。
Oehl et al. (2008)根据具孢子壁层结构、发芽盾特征、SSU和LSU序列分析,将盾巨孢囊霉属中的5个种分离出来建立了新属——盾孢囊霉属Cetraspora,吉尔莫盾孢囊霉C. gilmorei为模式种。目前该属包括杏黄盾孢囊霉C. armeniaca (Błaszkowski 1992)、吉尔莫盾孢囊霉C. gilmorei (Gerdemann & Trappe 1974)、透明盾孢囊霉C. pellucida (Nicolson & Schenck 1979)、条纹盾孢囊霉C. Striata (Cuenca & Herrera-Peraza 2008)、结节盾孢囊霉C. nodosa (Błaszkowski 1991)、瑞士盾孢囊霉C. helvetica (Oehl et al. 2010)和金黑盾孢囊霉C. auronigra (Lima et al. 2014)共7个物种(
本研究从贵州大学校内金佛山方竹根际土壤中分离到AM真菌的一个物种,经研究属于盾孢囊霉属中未被描述物种,对其进行了形态学描述和分子系统发育分析。
1 材料与方法
1.1 样品采集和AM真菌孢子分离
1.2 AM真菌单物种培养和离体培养
1.3 菌根结构染色
将单物种培养和离体培养所获得的根剪成0.5-1.0 cm,利用醋酸-墨水改良染色法(Vierheilig et al. 1998)进行染色。软化:将根样装入试管中,加入10%氢氧化钾溶液没过根样,90 ℃水浴10-15 min,至根样软化褪色,自来水缓慢冲洗,完全去除根样中的氢氧化钾。漂白:上述根样中加入20%的H2O2溶液,室温下静置30 min,自来水漂洗后,放置于新试管中。酸化:加入5%乙酸溶液酸化5 min,倒掉乙酸溶液。染色:加入含有5%醋酸-墨水染液(95 mL的5%醋酸+ 5 mL的Sheaffer Skrip蓝墨水),66 ℃水浴20- 30 min。脱色:倒掉染液,蒸馏水冲洗,根样浸泡在清水中进行褪色处理12 h,制片观察。
1.4 形态学分析
将湿筛法分离到的孢子去除表面杂质后,放置于载玻片上,以水或PVLG为载浮剂,在复合显微镜(Olympus BX53)和摄像系统(Olympus DP70)下观察、测量孢子颜色、大小、形状、孢子壁表面纹饰、层数、厚度、发芽壁结构,以及在Melzer’s试剂中的反应等特征,并进行拍照。参照国际丛枝菌根真菌保藏中心(INVAM,
1.5 DNA提取、PCR扩增及测序
表1 SSUmAf-LSUmAr与SSUmCf-LSUmBr引物信息
Table 1
| Nested-PCR | 引物 Primers | 序列 Sequence (5ʹ→3ʹ) | PCR产物长度 Length of PCR product (bp) |
|---|---|---|---|
| 第一轮扩增 First amplification | SSUmAf1 | TGGGTAATCTTTTGAAACTTYA | 1 800 |
| SSUmAf2 | TGGGTAATCTTRTGAAACTTCA | ||
| LSUmAr1 | GCTCACACTCAAATCTATCAAA | ||
| LSUmAr2 | GCTCTAACTCAATTCTATCGAT | ||
| LSUmAr3 | TGCTCTTACTCAAATCTATCAAA | ||
| LSUmAr4 | GCTCTTACTCAAACCTATCGA | ||
| 第二轮扩增 | SSUmCf1 | TCGCTCTTCAACGAGGAATC | 1 500 |
| Second amplification | SSUmCf2 | TATTGTTCTTCAACGAGGAATC | |
| SSUmCf3 | TATTGCTCTTNAACGAGGAATC | ||
| LSUmBr1 | DAACACTCGCATATATGTTAGA | ||
| LSUmBr2 | AACACTCGCACACATGTTAGA | ||
| LSUmBr3 | AACACTCGCATACATGTTAGA | ||
| LSUmBr4 | AAACACTCGCACATATGTTAGA | ||
| LSUmBr5 | AACACTCGCATATATGCTAGA |
1.6 序列比对和系统发育分析
将获得的正、反向序列利用SeqMan v. 7.0 (DNASTAR, Madison)进行校对和拼接,并将拼接序列提交到GenBank数据库中获得登录号。经BLAST比对后,下载盾孢囊霉属中具有SSU-ITS-LSU序列的全部物种及巨孢囊霉科其他属的物种共32个近缘种。以闪亮和平囊霉Pacispora scintillans (FM876831)为外群,进行最大似然法(ML)和贝叶斯法(BI)分析构建系统发
育树。需要说明的是目前盾孢囊霉属的7个物种中C. helvetica、C. Striata和C. auronigra无SSU-ITS-LSU序列。多基因片段数据矩阵通过MEGA 11软件中的MUSCLE功能进行校准。最大似然法(ML)分析是在CIPRES科学门户上使用RAxML-HPC2进行计算,选择GTRGAMMA模型,运行1 000次(Eduardo et al. 2020)。利用PhyloSuite v1.2.2软件中的ModeFinder选择最佳模型后(Zhang et al. 2020),使用Mrbayes 3.2进行贝叶斯系统发育分析。系统发育树在TreeGraph 2中进行查看与调整。
2 结果与分析
2.1 系统发育分析
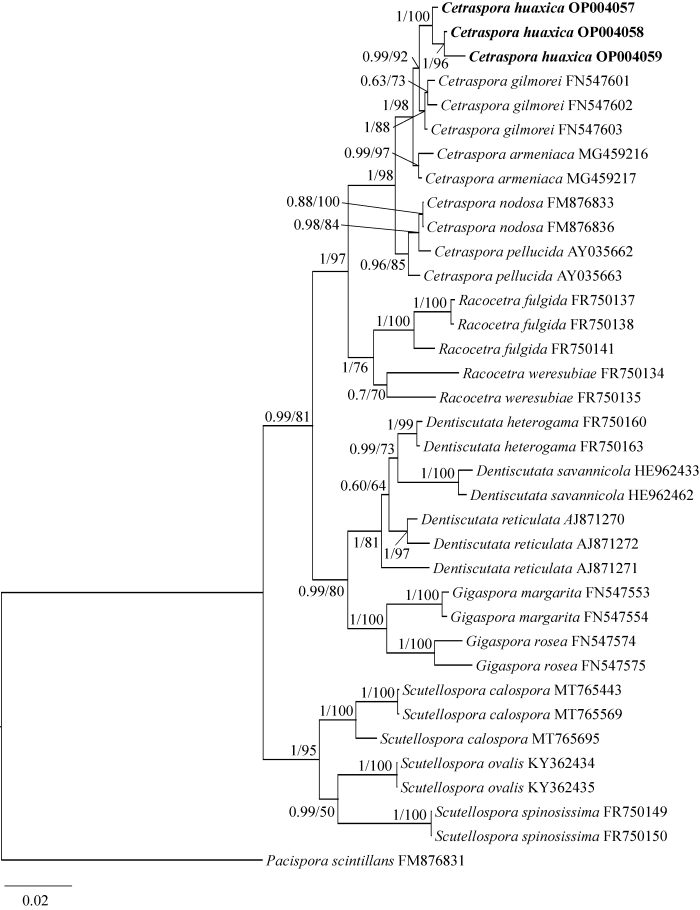
将测序所得的3条序列提交至NCBI GenBank数据库,获得登录号为OP004057、OP004058和OP004059,基于SSU-ITS-LSU序列,采用最大似然法(ML)和贝叶斯法(BI),以闪亮和平囊霉Pacispora scintillans (FM876831)为外群,构建系统发育树(图1)。结果显示:本研究物种属于盾孢囊霉属,3条序列以较高的支持率聚在一起形成独立分支,并与吉尔莫盾孢囊霉形成姊妹分支。
图1
图1
基于SSU-ITS-LSU序列的贝叶斯系统发育树 ML树的拓扑结构与贝叶斯分析结果相似,本图以贝叶斯拓扑结构展示;该系统发育树以闪亮和平囊霉Pacispora scintillans为外群;贝叶斯后验概率≥0.6及ML自举值≥50% (BYPP/MLBP)标注在节点位置;加粗标注为本研究分离的菌株;核苷酸替代率0.02
Fig. 1
Phylogenetic tree generated from Bayesian analysis based on SSU-ITS-LSU sequence, including Pacispora scintillans as outgroup. The tree topology of the maximum likelihood was similar to that of Bayesian analyses. Bayesian posterior probabilities ≥0.6 and ML bootstrap values ≥50% (BYPP/MLBP) are given above the nodes. The strains isolated in this study in bold; Scale in 0.02 substitution per site.
2.2 物种描述
花溪盾孢囊霉 新种 图2
Cetraspora huaxica D.Y. Chen, R.J. He & L. Jiang, sp. nov. Fig. 2
图2
图2
花溪盾孢囊霉形态特征 A-C:完整的孢子. D:PVLG中的破碎的孢子. 显示孢子壁层(OW和GW),发芽盾(gs),产孢细胞(Sc),萌发管(gt). E,F:Melzer’s试剂中孢子壁层结构(OW+MW+IW). IWL3与Melzer’s试剂反应呈深粉色至亮红棕色. G:产孢细胞壁层结构(ScWL1-2)及“菌丝钉”结构(peg). H-J:发芽盾上具有一个中央胚芽孔(gp),通常被大的折叠(f)隔开形成几个裂片,每个裂片上出现一个胚芽管起始点(gti). I,J:在PVLG溶液中用解剖针将发芽盾从孢子上剖离,且发芽盾壁(gsWL1-2)与Melzer’s试剂不发生反应. K,L:离体培养中的辅助细胞
Fig. 2
Microscopic morphological characteristics Cetraspora huaxica. A-C: Intact spores. D: Crushed spore on PVLG showing spore wall layers (OW and GW), germination shield (gs), a sporogenous cell (Sc), germinal tube (gt). E, F: In Melzer’s reagent, the spore wall layers (OW+MW+IW). IWL3 shows deep pink to bright red-brown reaction in Melzer’s reagent. G: Sporogenous cell wall layers (ScWL1-2) and ‘hyphal pegs’ structure. H-J: Germination shields (gs) with a initial central germ pore (gp), and several lobes that are generally separated by large folds (f); The lobes may regularly bear one germ tube initiation (gti). I, J: Independent germinated shields were completely separated from spores by tearing with a dissecting needle in PVLG liquid, and these shield wall layers (gsWL1-2) did not react in Melzer’s reagent. K, L: Auxiliary cells (aux) in root organ culture.
Fungal Name FN571069
Etymology: Huaxica (Latin), referring to Huaxi District, Guizhou Province, China, where this fungus was originally found.
Holotype: China. Guizhou Province: Huaxi District of Guiyang City (106°39ʹ31ʺE, 26°27ʹ13ʺN; 1 100-1 140 m above sea level), January 2022 and March 2022, D.Y. Chen, C.L. Long. Holotype, Fungarium of Insititute of Microbiology, CAS, voucher HMAS 286821. Isotypes were deposited at the Plant Physiology Laboratory of the University of Guizhou (GZ-HX-G01 to G07).
Description: Sporocarps unknown. Spores borne singly in soil, globose to subglobose, hyaline/white, 187-361×210-378 µm (wide× long), formed terminally on bulbous sporogenous cell (Figs. 2A-2C). The spore contents are hyaline and oil droplet shaped, and these oil droplets appear milky white near the germination shield as the spore maturates (Fig. 2B).
Subcellular structure of spores consists of an outer, middle and inner wall (Fig. 2D). Outer wall is composed of three layers. Outermost wall layer (OWL1) is hyaline, permanent, about 0.5-1.2 µm thick. OWL2 laminate and extremely rigid, hyaline, 3.80-8.76 µm thick, staining deep pink to bright red-brown in Melzer’s reagent (Figs. 2E, 2F). OWL3 tightly adherent to OWL2, 0.6-1.0 µm thick, difficult to observe in crushed spores.
Middle wall (MWL1-2) is 1.1-2.4 µm thick in total and two hyaline layers: a flexible outer layer MWL1 and a semi-flexible layer MWL2. MWL1 is 0.3-0.8 µm thick and generally does not separate from underlying MWL2 but often shows several folds in crushed spores. MWL2 is 0.8-1.6 µm thick, and generally more rigid than MWL1. None of the layers reacts to Melzer’s (Fig. 2E).
Inner wall (IW) is triple-layered (Fig. 2E). The outer IW layer (IWL1) is hyaline, semi-flexible and 0.5-1.4 μm thick. The second layer (IWL2) is semi-flexible, and is 0.6-1.2 µm thick. The innermost layer (IWL3) is rigid, mostly tightly adherent to IWL2. IWL3 stains deep pink to bright red-brown when exposed to Melzer’s reagent.
Germination shield forms on the outer IW surface, pale yellow to pale yellow brown, ellipsoid to cardioid, 88.6-126.1×113.3-147.9 µm wide × long, partitioning 4-8 lobes with nicked margins formed by shallow incisions. Generally, each lobe had a circular germ tube origin (gti; 4.2-10.3 µm in diam.) (Figs. 2H-2J).
Sporogenous cell (Sc) is globose to elongate, 32-49 µm wide and generally pale yellow to pale yellow brown. Two wall layers are visible on the young sporogenous cell, which are continuous with OWL1 and with laminated OWL2. Layer 1 is hyaline, 0.4-0.9 µm thick. Layer 2 is pale yellow to pale yellow brown, 1.2-2.6 µm thick at the sporebase, thinning distally to about 1 µm. One to two (rarely) ‘hyphal pegs’ are often formed on the sporogenous cells (Figs. 2C, 2G).
Auxiliary cells (aux) are formed in small aggregates (2-8 cells) on coiled hyaline hyphae 3.5-5.5 μm in diameter. Each cell (14.80-28.35× 21.20-30.65 μm, wide × long) in aggregates is subglobose, pale-yellow, cells almost smooth or ornamented with tuberculate surface, with swellings 1-6 μm high and 3-8 μm wide (Figs. 2K, 2L).
Mycorrhizal associations. By dual culture and in single-species cultures with Trifolium repens L. as host plant, C. huaxica formed mycorrhiza with vesicles, intra- and extra-radical hyphae (Fig. 3).
Distribution and habitat: Spores of C. huaxica were isolated from rhizosphere soil of Chimonobambusa utilis in Huaxi District, Guizhou Province, China where had typical subtropical humid mild climate, with no severe cold in winter and no heat in summer. It has a long frost-free period (The average frost-free period is 246 d) and abundant rainfall (the annual rainfall is 1 178.3 mm) and high humidity (the mean annual temperature was about 14.9 ℃). Soils are yellowish soil, with 2.7% of organic matter and a pH ranging from 4.5-5.5. At the same time, this site is the only place where this fungus has been found so far.
词源:huaxica (拉丁语),指中国贵州省花溪区,该真菌最初被发现的地方。
主模式标本:贵州省贵阳市花溪区(东经106°39′31ʺ,北纬26°27′13ʺ,2022年1月、 2022年3月,陈德瑶,龙春丽。模式标本存于中国科学院微生物研究所菌物标本馆(主模标本:HMAS 286821;副模标本:HMAS 286822-23)。同型标本保存于贵州大学植物生理实验室(GZ-HX-G01-G07)。
中壁层由2层(MWL1-2),透明,厚度约为1.1-2.4 µm,MWL1为柔性壁,厚约0.3-0.8 µm,通常不与底层的MWL2分开,仅在压碎后的孢子中出现几条褶皱;MWL2为半柔性壁层,厚约0.8-1.6 µm,通常比MWL1坚硬。MWL1-2在Melzer’s试剂中不发生反应(图2E)。
内壁层3层(IWL1-3)。IWL1透明,半柔性壁,厚0.5-1.4 μm;IWL2半柔性壁,厚0.6-1.2 µm;IWL3为刚性壁,与IWL2紧密贴合,在Melzer’s试剂中染色为深粉色至亮红棕色。
发芽盾(gs)位于内壁层表面,颜色呈浅黄色至淡黄棕色,椭圆形至心形,大小88.6-126.1× 113.3-147.9 µm,具有4-8个裂片,裂片边缘有刻痕。每个裂片通常有一个圆形胚芽管起始点(gti),直径4.2-10.3 μm (图2H-2J)。
产孢细胞(Sc)近球形至棒状,宽约32-49 µm,通常为浅黄色至淡黄棕色。具有2层壁组成(ScWL1-2),分别与外壁层OWL1和OWL2相连。ScWL1透明,厚0.4-0.9 µm;ScWL2为浅黄色至淡黄棕色,孢子基部厚1.2-2.6 µm,远端变薄至约1 µm。通常在产孢细胞上形成1-2个(偶见)菌丝钉(peg) (图2C,2G)。
辅助细胞(aux)通常2-8个聚集成簇,着生于直径为3.5-5.5 μm的卷曲透明的菌丝上,近球形,细胞大小14.80-28.35×21.20-30.65 μm,细胞壁淡黄色,通常表面光滑或有瘤状突起,突起高1-6 μm,宽3-8 μm (图2K,2L)。
菌根染色:通过单物种培养和离体培养得到的菌根,经染色后观察到泡囊、根内和根外菌丝结构(图3)。
图3
图3
花溪盾孢囊霉菌根结构 A:离体培养中的菌丝网络和辅助细胞. B,C:利用0.5%醋酸墨水染色后的三叶草菌根图(菌丝和泡囊结构)
Fig. 3
Mycorrhizal structures of Cetraspora huaxica. A: Hyphal network and auxiliary cells in dual culture. B, C: Mycorrhizal structures of C. huaxica in roots of Trifolium repens L. stained in 0.5% ink-vinegar (hyphae and vesicles).
分布与生境:该物种是从贵州省花溪地区金佛山方竹根际土壤中分离得到。该地区(东经106°39′31ʺ,北纬26°27′13ʺ;海拔1 100-1 140 m)为典型的亚热带湿润温和型气候,年平均气温约14.9 ℃,平均无霜期246 d,年降雨量1 178.3 mm。土壤类型为黄壤,有机质含量为2.7%,pH值在4.5-5.5。这是迄今为止该物种的唯一发现地。
2.3 盾孢囊霉属Cetraspora物种检索表
本检索表在Oehl et al. (2008)的基础上进行更新与修正,表中共8个种。
1 孢子壁表面无纹饰 2
1 孢子壁表面有纹饰 3
2 孢子透明至白色 4
2 孢子有颜色 5
3 孢子颜色较淡,且孢子直径大小约250 μm左右 6
3 孢子呈微带粉红的黄褐色,直径110-190 μm,孢子表面有指纹状突起 条纹盾孢囊霉C. striata
4 孢子明亮透明,白色至浅灰色,一般呈球形到近球形,直径(60)120-250(-420) μm;产孢细胞透明至半透明,发芽盾一般不可见 透明盾孢囊霉C. pellucid
4 孢子透明,产孢细胞和发芽盾易观察 7
5 孢子杏黄色到黄棕色,直径140-240 μm 杏黄盾孢囊霉C. armeniaca
5 孢子亮黄色至金黄色,孢子内壁层L2在Melzer’s试剂中反应变为深紫色至紫黑色 金黑盾孢囊霉C. auronigra
6 幼嫩孢子呈亮白色,在土壤中老化后变暗至乳白色,孢子外壁有凸疣,外壁层OWL2-3和内壁层IWL2在Melzer’s试剂中反应呈深紫色到黑紫色,直径210-270 µm 瑞士盾孢囊霉C. helvetica
6 孢子透明至淡黄色,在孢子外壁上有结节状突起;直径大小为160-270 μm 结节盾孢囊霉C. nodosa
7 孢子透明,在福尔马林中呈奶油色,产孢细胞为棕色;发芽盾易观察,球形至近球形,直径200-320 μm,孢子壁层为3层(外壁层:OWL1-2, 中壁层:MWL1-2,内壁层:IWL1-2) 吉尔莫盾孢囊霉C. gilmorei
7 孢子透明,产孢细胞呈淡黄褐色,发芽盾呈浅黄色到淡黄棕色易观察,孢子外壁层(OWL1-3)和内壁层(IWL1-3)都有3层,且OWL2和IWL3在Melzer’s试剂中呈深粉红色到亮红棕色。孢子大小为187-361×210-378 μm 花溪盾孢囊霉C. huaxica
3 讨论
盾孢囊霉属物种的形态特征:孢子具有3个孢子壁层、发芽盾多裂,具4-12个裂片,孢子透明至近透明(Oehl et al. 2008),花溪盾孢囊霉与以上特征吻合,鉴定为盾孢囊霉属,其内壁层3层(IWL1-3),IWL3在Melzer’s试剂中呈深粉红色到亮红棕色,易于与本属其他已描述的物种区分。
在形态学上,花溪盾孢囊霉与透明盾孢囊霉、吉尔莫盾孢囊霉的孢子关系密切,孢子均透明至近透明,孢子都较大,通常在200-280 μm,但孢子壁层和Melzer’s试剂中染色特征具有明显差异。透明盾孢囊霉与花溪盾孢囊霉孢子均为无色,内含物呈油滴状,但透明盾孢囊霉发芽盾不可见,孢子内壁层仅有2层,IWL2在Melzer’s试剂中呈现深紫色到紫黑色(Gerdemann & Trappe 1974;Nicolson & Schenck 1979;Oehl et al. 2008);而花溪盾孢囊霉发芽盾呈淡黄棕色易观察,孢子内壁层为3层,IWL3在Melzer’s试剂中呈现出深粉色到亮红棕色(图2E)。自然条件下花溪盾孢囊霉与吉尔莫盾孢囊霉孢子形状、颜色相近,但孢子壁结构存在差异。吉尔莫盾孢囊霉孢子外壁(OW)和内壁(IW)均为2层,而花溪盾孢囊霉孢子外壁(OW)和内壁(IW)均有3层。在Melzer’s试剂中,吉尔莫盾孢囊霉OWL2和IWL2染色均为红棕色(Gerdemann & Trappe 1974;Oehl et al. 2008);花溪盾孢囊霉OWL2染色为亮红棕色,IWL3染色为深粉色到亮红棕色,IWL2在Melzer’s试剂中不发生反应。
综上所述,结合形态学与系统发育分析我们得出结论,本研究描述的物种为AM真菌的一个新种,迄今为止仅在贵阳市花溪区的金佛山方竹的根际土壤中发现,但该物种是否在其他地区及生境分布,还需进一步实地研究。中国地域辽阔,植被复杂多样,拥有超过3万种植物,AM 真菌物种资源潜力巨大。目前在中国发现并报道了包括17个新种在内的AM真菌共有158种(王幼珊和刘润进 2017;姚莉梅等 2019, 2020;He et al. 2021;朱青青等 2021;Long et al. 2022;Yu et al. 2022),不到全球已报道AM真菌种数的一半,显然,我国AM真菌物种数量(新种)被严重低估,更多的AM真菌物种正在等待被发现。
致谢
感谢王艳和龙青在物种收集和鉴定及试验中的帮助,同时感谢苟光前教授(贵州大学)对新物种拉丁名称方面的指导。
参考文献
Gigaspora margarita, a new species in the Endogonaceae
Structure and hyperparasitism of a new species of Gigaspora
DOI:10.1016/S0007-1536(82)80096-X URL [本文引用: 1]
Polish Glomales Ⅷ. Scutellospora nodosa, a new species with knobby spores
DOI:10.1080/00275514.1991.12026048 URL [本文引用: 1]
Scutellospora armeniaca, a new species in Glomales (Zygomycetes) from Poland
DOI:10.1080/00275514.1992.12026232 URL [本文引用: 2]
The arbuscular mycorrhizal Paraglomus majewskii sp. nov. represents a new distinct basal lineage in Paraglomeraceae (Glomeromycota)
DOI:10.3852/10-430 URL [本文引用: 1]
Scutellospora striata sp. nov., a newly described glomeromycotan fungus from La Gran Sabana, Venezuela
Dual axenic culture of sheared-root inocula of vesicular-arbuscular mycorrhizal fungi associated with tomato roots
DOI:10.1007/BF00204015 URL [本文引用: 1]
Dual culture of tobacco Va 116 hairy roots and Acaulospora koskei
Septoglomus mexicanum, a new species of arbuscular mycorrhizal fungi from semiarid regions in Mexico
DOI:10.1080/00275514.2019.1689748 URL [本文引用: 1]
Spores of mycorrhizal Endogone species extracted from soil by wet sieving and decanting
The Endogonaceae in the Pacific North West
Acaulospora fanjing (Glomeromycota), a new species of arbuscular mycorrhizal fungi from Fanjingshan in China
Diversity of arbuscular mycorrhizal fungi in mossy dwarf forest of Fanjing Mountain, Guizhou Province, Southwest China
Production of Glomus intraradices propagules, an arbuscular mycorrhizal fungus, in an airlift bioreactor
A history of research on arbuscular mycorrhiza
This is not a review paper in the traditional sense, of which there are many. Three of the most influential reviews that summarized well some of the "older" literature include those by Nicolson (1967), Gerdemann (1968) and Mosse (1973). Instead, in this brief and incomplete work, we attempt to show the historical development of research on arbuscular mycorrhizas. We owe much to those who have written other historical accounts, including Rayner (1926-1927), Trappe and Berch (1985), Mosse (1985), Schenck (1985), Harley (1991) and Allen (1996), but the contents of this work naturally reflect our own ignorance, interests and biases. It was often difficult to distinguish between the historical and the contemporary, and we did not use any specific cutoff date in making this distinction. The degree to which we include "contemporary" literature was determined by our own assessment of its connectedness to older literature. In any case, we hope this will be of some interest to those of you who study the arbuscular mycorrhiza, and that it will serve the purpose of providing what we consider to be an important historical context for current researchers. We wish you good fortune in your research.
The role of in vitro cultivation on asymbiotic trait variation in a single species of arbuscular mycorrhizal fungus
DOI:S1878-6146(18)30295-2
PMID:30928039
[本文引用: 1]

Cultivating arbuscular mycorrhizal (AM) fungi in vitro is an efficient way to produce material for industry and research. However, such artificial growing conditions may impose selective pressure on fungi grown in vitro over many generations. We hypothesized that isolates subjected to long term propagation in vitro may develop increasingly ruderal traits. We proposed a predictive framework for the effect of in vitro cultivation on asymbiotic AM fungal traits. Using photomicrography and image processing, we analyzed morphology and growth traits for 14 isolates representing an in vitro cultivation gradient from 0 to >80 generations in vitro. We investigated the range of trait variation among asymbiotic growth of arbuscular mycorrhizal (AM) fungus isolates (Rhizoglomus irregulare). Spore dormancy was strongly associated with in vitro cultivation. We observed extremely high levels of inter-isolate variation for most fungal traits, but this was not related to time in vitro. Our results indicate that intra-specific diversity may have a strong ecological role in AM fungal communities.Copyright © 2019 British Mycological Society. Published by Elsevier Ltd. All rights reserved.
Gigaspora erythropa, a new species forming arbuscular mycorrhizae
DOI:10.1080/00275514.1984.12023833 URL [本文引用: 1]
Species of Gigaspora (Endogonaceae) with roughened walls
DOI:10.1080/00275514.1985.12025159 URL [本文引用: 1]
Phylogenetic reference data for systematics and phylotaxonomy of arbuscular mycorrhizal fungi from phylum to species level
DOI:10.1111/j.1469-8137.2011.03962.x
PMID:22150759
[本文引用: 1]

Although the molecular phylogeny, evolution and biodiversity of arbuscular mycorrhizal fungi (AMF) are becoming clearer, phylotaxonomically reliable sequence data are still limited. To fill this gap, a data set allowing resolution and environmental tracing across all taxonomic levels is provided. Two overlapping nuclear DNA regions, totalling c. 3 kb, were analysed: the small subunit (SSU) rRNA gene (up to 1800 bp) and a fragment spanning c. 250 bp of the SSU rDNA, the internal transcribed spacer (ITS) region (c. 475-520 bp) and c. 800 bp of the large subunit (LSU) rRNA gene. Both DNA regions together could be analysed for 35 described species, the SSU rDNA for c. 76 named and 18 as yet undefined species, and the ITS region or LSU rDNA, or a combination of both, for c. 91 named and 16 as yet undefined species. Present phylogenetic analyses, based on the three rDNA markers, provide reliable and robust resolution from phylum to species level. Altogether, 109 named species and 27 cultures representing as yet undefined species were analysed. This study provides a reference data set for molecular systematics and environmental community analyses of AMF, including analyses based on deep sequencing.© 2011 The Authors. New Phytologist © 2011 New Phytologist Trust.
Cetraspora auronigra, a new glomeromycete species from Ouro Preto (Minas Gerais, Brazil)
Gigaspora polymorphira, a new species of arbuscular mycorrhizal fungi (Glomeromycota) from Fanjingshan National Nature Reserve in China
DOI:10.11646/phytotaxa.572.2 URL [本文引用: 1]
Revised classification of arbuscular mycorrhizal fungi (Zygomycetes): a new order, Glomales, two new suborders, Glomineae and Gigasporinae, and two families, Acaulosporaceae and Gigasporaceae, with an emendation of Glomaceae
Endogonaceous mycorrhizal endophytes in Florida
DOI:10.1080/00275514.1979.12020997 URL [本文引用: 2]
Revision of Scutellospora and description of five new genera and three new families in the arbuscular mycorrhiza-forming Glomeromycetes
Cetraspora helvetica, a new ornamented species in the Glomeromycetes from Swiss agricultural fields
DOI:10.5248/114.71 URL [本文引用: 1]
Optimization on the conditions of AM fungal secondary sporulation in the dual vitro culture
Arbuscular mycorrhizal fungi communities from tropical Africa reveal strong ecological structure
DOI:10.1111/nph.14122
PMID:27560189
[本文引用: 1]

Understanding the distribution and diversity of arbuscular mycorrhizal fungi (AMF) and the rules that govern AMF assemblages has been hampered by a lack of data from natural ecosystems. In addition, the current knowledge on AMF diversity is biased towards temperate ecosystems, whereas little is known about other habitats such as dry tropical ecosystems. We explored the diversity and structure of AMF communities in grasslands, savannas, dry forests and miombo in a protected area under dry tropical climate (Gorongosa National Park, Mozambique) using 454 pyrosequencing. In total, 147 AMF virtual taxa (VT) were detected, including 22 VT new to science. We found a high turnover of AMF with ˂ 12% of VT present in all vegetation types. Forested areas supported more diverse AMF communities than savannas and grassland. Miombo woodlands had the highest AMF richness, number of novel VT, and number of exclusive and indicator taxa. Our data reveal a sharp differentiation of AMF communities between forested areas and periodically flooded savannas and grasslands. This marked ecological structure of AMF communities provides the first comprehensive landscape-scale evidence that, at the background of globally low endemism of AMF, local communities are shaped by regional processes including environmental filtering by edaphic properties and natural disturbance.© 2016 The Authors. New Phytologist © 2016 New Phytologist Trust.
Nomenclatural clarifications and new taxa in the Glomeromycota
DOI:10.1017/S0953756204231173 URL [本文引用: 1]
Mycorrhizal ecology and evolution: the past, the present, and the future
DOI:10.1111/nph.13288
PMID:25639293
[本文引用: 1]

Almost all land plants form symbiotic associations with mycorrhizal fungi. These below-ground fungi play a key role in terrestrial ecosystems as they regulate nutrient and carbon cycles, and influence soil structure and ecosystem multifunctionality. Up to 80% of plant N and P is provided by mycorrhizal fungi and many plant species depend on these symbionts for growth and survival. Estimates suggest that there are c. 50 000 fungal species that form mycorrhizal associations with c. 250 000 plant species. The development of high-throughput molecular tools has helped us to better understand the biology, evolution, and biodiversity of mycorrhizal associations. Nuclear genome assemblies and gene annotations of 33 mycorrhizal fungal species are now available providing fascinating opportunities to deepen our understanding of the mycorrhizal lifestyle, the metabolic capabilities of these plant symbionts, the molecular dialogue between symbionts, and evolutionary adaptations across a range of mycorrhizal associations. Large-scale molecular surveys have provided novel insights into the diversity, spatial and temporal dynamics of mycorrhizal fungal communities. At the ecological level, network theory makes it possible to analyze interactions between plant-fungal partners as complex underground multi-species networks. Our analysis suggests that nestedness, modularity and specificity of mycorrhizal networks vary and depend on mycorrhizal type. Mechanistic models explaining partner choice, resource exchange, and coevolution in mycorrhizal associations have been developed and are being tested. This review ends with major frontiers for further research.© 2015 The Authors. New Phytologist © 2015 New Phytologist Trust.
Ink and vinegar, a simple staining technique for arbuscular- mycorrhizal fungi
DOI:10.1128/AEM.64.12.5004-5007.1998
PMID:9835596
[本文引用: 1]

We developed a reliable, inexpensive, and simple method for staining arbuscular-mycorrhizal fungal colonizations in root tissues. Apart from applications in research, this nontoxic, high-quality staining method also could be of great utility in teaching exercises. After adequate clearing with KOH, an ink-vinegar solution successfully stained all fungal structures, rendering them clearly visible.
Taxonomic concepts in the Endogonaceae: Ⅲ. The separation of Scutellospora gen. nov. from Gigaspora Gerd. & Trappe
Nutrient exchange and regulation in arbuscular mycorrhizal symbiosis
DOI:S1674-2052(17)30228-9
PMID:28782719
[本文引用: 1]

Most land plants form symbiotic associations with arbuscular mycorrhizal (AM) fungi. These are the most common and widespread terrestrial plant symbioses, which have a global impact on plant mineral nutrition. The establishment of AM symbiosis involves recognition of the two partners and bidirectional transport of different mineral and carbon nutrients through the symbiotic interfaces within the host root cells. Intriguingly, recent discoveries have highlighted that lipids are transferred from the plant host to AM fungus as a major carbon source. In this review, we discuss the transporter-mediated transfer of carbon, nitrogen, phosphate, potassium and sulfate, and present hypotheses pertaining to the potential regulatory mechanisms of nutrient exchange in AM symbiosis. Current challenges and future perspectives on AM symbiosis research are also discussed.Copyright © 2017 The Author. Published by Elsevier Inc. All rights reserved.
A checklist of arbuscular mycorrhizal fungi in the recent taxonomic system of Glomeromycota
Dual culture of tobacco hairy roots and AM fungus of Gigaspora
Three new records of arbuscular mycorrhizal fungi from Fanjing Mountain in China
Acaulospora brasiliensis, a new Chinese record species of Glomeromycota through morphology and molecular phylogeny identification
Glomus chinense and Dominikia gansuensis, two new Glomeraceae species of arbuscular mycorrhizal fungi from high altitude in the Tibetan Plateau
DOI:10.1007/s11557-022-01774-4 [本文引用: 1]
PhyloSuite: an integrated and scalable desktop platform for streamlined molecular sequence data management and evolutionary phylogenetics studies
DOI:10.1111/1755-0998.13096
PMID:31599058
[本文引用: 1]

Multigene and genomic data sets have become commonplace in the field of phylogenetics, but many existing tools are not designed for such data sets, which often makes the analysis time-consuming and tedious. Here, we present PhyloSuite, a (cross-platform, open-source, stand-alone Python graphical user interface) user-friendly workflow desktop platform dedicated to streamlining molecular sequence data management and evolutionary phylogenetics studies. It uses a plugin-based system that integrates several phylogenetic and bioinformatic tools, thereby streamlining the entire procedure, from data acquisition to phylogenetic tree annotation (in combination with iTOL). It has the following features: (a) point-and-click and drag-and-drop graphical user interface; (b) a workplace to manage and organize molecular sequence data and results of analyses; (c) GenBank entry extraction and comparative statistics; and (d) a phylogenetic workflow with batch processing capability, comprising sequence alignment (mafft and macse), alignment optimization (trimAl, HmmCleaner and Gblocks), data set concatenation, best partitioning scheme and best evolutionary model selection (PartitionFinder and modelfinder), and phylogenetic inference (MrBayes and iq-tree). PhyloSuite is designed for both beginners and experienced researchers, allowing the former to quick-start their way into phylogenetic analysis, and the latter to conduct, store and manage their work in a streamlined way, and spend more time investigating scientific questions instead of wasting it on transferring files from one software program to another.© 2019 John Wiley & Sons Ltd.
Three new recorded species of AM fungi from Fanjing Mountain in China
中国新记录种:巴西无梗囊霉的形态与分子生物学鉴定